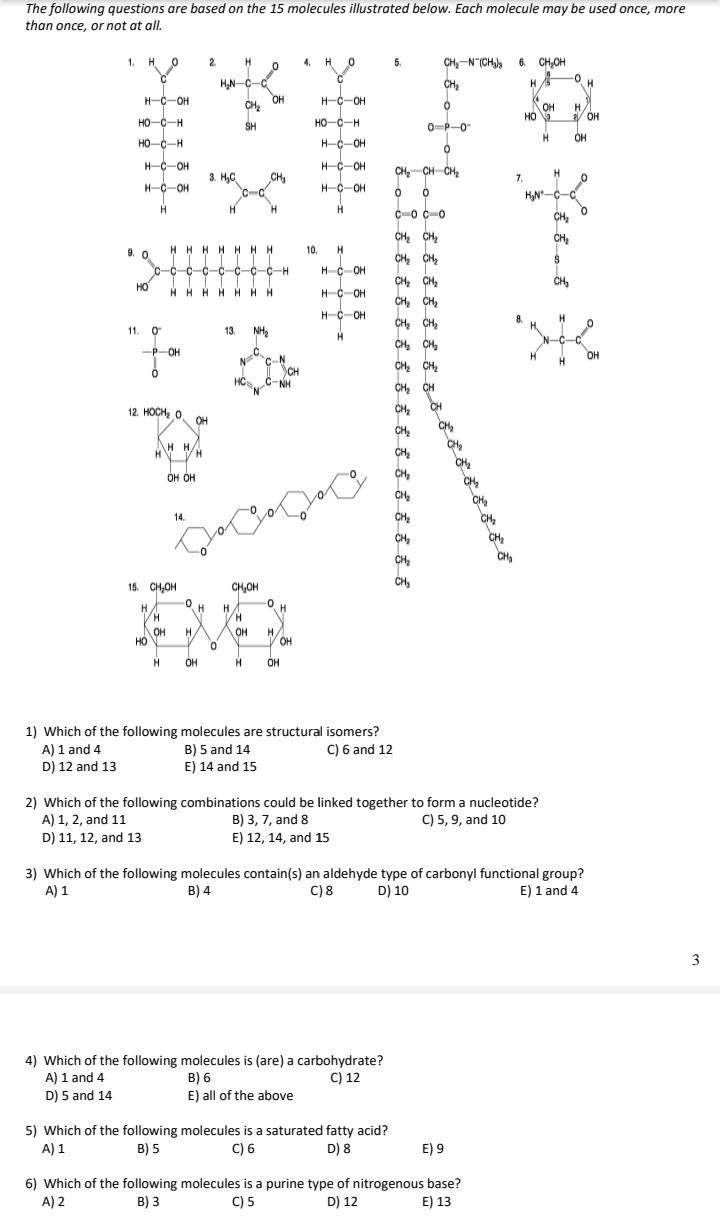
The following questions are based on the 15 molecules illustrated below. Each molecule may be used...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
The following questions are based on the 15 molecules illustrated below. Each molecule may be used once, more than once, or not at all. 1. H O 2 4. H O 5. CH,-N (CH, 6 CH,OH CH 0- HN-C-C OH CH H. H-C-OH H-C-OH OH / OH HO-C-H SH HO-C-H 0-P-0 HO-C-H HC-OH H. OH H-C-OH H-C-OH CH, CH CH, 3. H,C CH 7. H-C-OH HC-OH HN-C H H. H. C-o C-0 CH, CH CH2 CH, HHHHHHH 9. 0 c-c-c-c-c-c-c-c-H 10. CH CH2 HC-OH CH CH, CH, CH, CH, CH, CH, HO CH HHHH HHH HC-OH H-C-OH H. 11. 0 13. NH2 CH, CH, CH, CH, CH -P-OH C. N C-N CH C-NH OH 12. HOCH, O CH, CH OH CH, H H/ CH, CH, CH, OH OH CH CH, CH, CH, 14. CH, CH, CH 15. CH,OH CH,OH H. H. H OH HO H. H. V OH OH OH H. OH 1) Which of the following molecules are structural isomers? B) 5 and 14 E) 14 and 15 A) 1 and 4 D) 12 and 13 C) 6 and 12 2) Which of the following combinations could be linked together to form a nucleotide? B) 3, 7, and 8 E) 12, 14, and 15 A) 1, 2, and 11 C) 5, 9, and 10 D) 11, 12, and 13 3) Which of the following molecules contain(s) an aldehyde type of carbonyl functional group? C) 8 A) 1 B) 4 D) 10 E) 1 and 4 4) Which of the following molecules is (are) a carbohydrate? B) 6 E) all of the above C) 12 A) 1 and 4 D) 5 and 14 5) Which of the following molecules is a saturated fatty acid? C) 6 A) 1 B) 5 D) 8 E) 9 6) Which of the following molecules is a purine type of nitrogenous base? C) 5 A) 2 B) 3 D) 12 E) 13 7) Which of the following molecules act as building blocks (monomers) of polypeptides? A) 1, 4, and 6 C) 7, 8, and 13 D) 11, 12, and 13 B) 2, 7, and 8 E) 12, 13, and 15 8) Which of the following molecules is an amino acid with a hydrophobic R group or side chain? A) 3 C) 7 B) 5 D) 8 E) 12 9) Which of the following molecules could be joined together by a peptide bond as a result of a dehydration reaction? A) 2 and 3 B) 3 and 7 C) 7 and 8 D) 8 and 9 E) 12 and 13 10) A fat (or triacylglycerol) would be formed as a result of a dehydration reaction between A) one molecule of 9 and three molecules of 10. B) three molecules of 9 and one molecule of 10. C) one molecule of 5 and three molecules of 9. D) three molecules of 5 and one molecule of 9. E) one molecule of 5 and three molecules of 10. 11) Which of the following molecules could be joined together by a phosphodiester type of covalent bond? A) 3 and 4 D) 11 and 12 C) 6 and 15 B) 3 and 8 E) 11 and 13 12) Which of the following molecules is the pentose sugar found in RNA? A) 1 B) 4 C) 6 D) 12 E) 13 13) Which of the following molecules contains a glycosidic linkage type of covalent bond? A) 4 C) 12 B) 6 D) 13 E) 15 14) Which of the following molecules has (have) a functional group that frequently is involved in maintaining th tertiary structure of a protein? A) 2 B) 3 C) 9 D) 11 E) 9 and 11 15) Which of the following molecules consists of a hydrophilic "head" region and a hydrophobic "tail" region? A) 2 B) 5 C) 7 D) 9 E) 11 16) Which of the following statements is false? A) 1 and 4 could be joined together by a glycosidic linkage to form a disaccharide. B) 9 and 10 could be joined together by ester bonds to form a triacylglycerol. C) 2 and 7 could be joined together to form a short peptide. D) 2, 7, and 8 could be joined together to form a short peptide. E) 14 and 15 could be joined together to form a polypeptide. The following questions are based on the 15 molecules illustrated below. Each molecule may be used once, more than once, or not at all. 1. H O 2 4. H O 5. CH,-N (CH, 6 CH,OH CH 0- HN-C-C OH CH H. H-C-OH H-C-OH OH / OH HO-C-H SH HO-C-H 0-P-0 HO-C-H HC-OH H. OH H-C-OH H-C-OH CH, CH CH, 3. H,C CH 7. H-C-OH HC-OH HN-C H H. H. C-o C-0 CH, CH CH2 CH, HHHHHHH 9. 0 c-c-c-c-c-c-c-c-H 10. CH CH2 HC-OH CH CH, CH, CH, CH, CH, CH, HO CH HHHH HHH HC-OH H-C-OH H. 11. 0 13. NH2 CH, CH, CH, CH, CH -P-OH C. N C-N CH C-NH OH 12. HOCH, O CH, CH OH CH, H H/ CH, CH, CH, OH OH CH CH, CH, CH, 14. CH, CH, CH 15. CH,OH CH,OH H. H. H OH HO H. H. V OH OH OH H. OH 1) Which of the following molecules are structural isomers? B) 5 and 14 E) 14 and 15 A) 1 and 4 D) 12 and 13 C) 6 and 12 2) Which of the following combinations could be linked together to form a nucleotide? B) 3, 7, and 8 E) 12, 14, and 15 A) 1, 2, and 11 C) 5, 9, and 10 D) 11, 12, and 13 3) Which of the following molecules contain(s) an aldehyde type of carbonyl functional group? C) 8 A) 1 B) 4 D) 10 E) 1 and 4 4) Which of the following molecules is (are) a carbohydrate? B) 6 E) all of the above C) 12 A) 1 and 4 D) 5 and 14 5) Which of the following molecules is a saturated fatty acid? C) 6 A) 1 B) 5 D) 8 E) 9 6) Which of the following molecules is a purine type of nitrogenous base? C) 5 A) 2 B) 3 D) 12 E) 13 7) Which of the following molecules act as building blocks (monomers) of polypeptides? A) 1, 4, and 6 C) 7, 8, and 13 D) 11, 12, and 13 B) 2, 7, and 8 E) 12, 13, and 15 8) Which of the following molecules is an amino acid with a hydrophobic R group or side chain? A) 3 C) 7 B) 5 D) 8 E) 12 9) Which of the following molecules could be joined together by a peptide bond as a result of a dehydration reaction? A) 2 and 3 B) 3 and 7 C) 7 and 8 D) 8 and 9 E) 12 and 13 10) A fat (or triacylglycerol) would be formed as a result of a dehydration reaction between A) one molecule of 9 and three molecules of 10. B) three molecules of 9 and one molecule of 10. C) one molecule of 5 and three molecules of 9. D) three molecules of 5 and one molecule of 9. E) one molecule of 5 and three molecules of 10. 11) Which of the following molecules could be joined together by a phosphodiester type of covalent bond? A) 3 and 4 D) 11 and 12 C) 6 and 15 B) 3 and 8 E) 11 and 13 12) Which of the following molecules is the pentose sugar found in RNA? A) 1 B) 4 C) 6 D) 12 E) 13 13) Which of the following molecules contains a glycosidic linkage type of covalent bond? A) 4 C) 12 B) 6 D) 13 E) 15 14) Which of the following molecules has (have) a functional group that frequently is involved in maintaining th tertiary structure of a protein? A) 2 B) 3 C) 9 D) 11 E) 9 and 11 15) Which of the following molecules consists of a hydrophilic "head" region and a hydrophobic "tail" region? A) 2 B) 5 C) 7 D) 9 E) 11 16) Which of the following statements is false? A) 1 and 4 could be joined together by a glycosidic linkage to form a disaccharide. B) 9 and 10 could be joined together by ester bonds to form a triacylglycerol. C) 2 and 7 could be joined together to form a short peptide. D) 2, 7, and 8 could be joined together to form a short peptide. E) 14 and 15 could be joined together to form a polypeptide.
Expert Answer:
Answer rating: 100% (QA)
7 Molecule 4is the structure of D Glucose and CA Epimor change at a Hace C4 at... View the full answer

Related Book For 

Intermediate accounting
ISBN: 978-0077647094
7th edition
Authors: J. David Spiceland, James Sepe, Mark Nelson
Posted Date:
Students also viewed these chemical engineering questions
-
The following questions are adapted from a variety of sources including questions developed by the AICPA Board of Examiners and those used in the Kaplan CPA Review Course to study property, plant,...
-
The following questions are adapted from a variety of sources including questions developed by the AICPA Board of Examiners and those used in the Kaplan CPA Review Course to study property, plant,...
-
The following questions are part of an internal control questionnaire for the acquisition and expenditure process. The client has been asked to answer the questions for the acquisition and...
-
Problem 5-47 Amortizing Loans and Inflation (LO3) Suppose you take out a $108,000, 20-year mortgage loan to buy a condo. The interest rate on the loan is 5%. To keep things simple, we will assume you...
-
In a CMO structure with several PAC bonds, explain why, when the support bonds are paid off, the structure will be just like a sequential-pay CMO.
-
A passenger car weighs 20.02 kN and has a wheelbase of 2.79 m. The center of gravity is 1.27 m behind the front axle. If a pair of radial-ply tires with cornering stiffness of 45.88 kN/rad, are...
-
Top managers of Bernard Associates are reviewing company perfor- mance for 2009. The income statement reports a 15% increase in net income, which is outstanding. The balance sheet shows modest...
-
After five years with a national CPA firm with mostly large manufacturing clients, Amy Kimbell joined Hi-Quality Productions Inc. (Hi-Q) as manager of Manufacturing Accounting. Amy has both CPA and...
-
For Libell & Son decisions constantly must be made. In the wholesale business, each customer you deal with and each market you are in will differ. What you establish as the protocol to correct a...
-
The proposed rates were not in the range the CEO expected given the pricing analysis. The CEO has asked the pricing actuary to verify the total projected loss cost excluding potential large storm...
-
Suppose the spot ask exchange rate, Sa($|), is $1.21 = 1.00 and the spot bid exchange rate, Sb($|), is $1.20 = 1.00. If you were to buy $10,000,000 worth of British pounds and then sell them five...
-
Marty, age 56, and Marcia, age 53, are starting to think about retirement. Marty plans to retire at age 65 and he expects to live to age 85. Marcia plans to retire at age 62 and she expects to live...
-
Solve the equation -16x + 4y = 32 for y.
-
Two blocks of masses m = 14, 1 kg and m : 7, 3 kg are connected by a rope that hangs over a pulley as shown in the figure. The pulley is a uniform disk of radius R = 0, 22 m and the mass M = 5 kg....
-
According to the ideas behind the revenue recognition standard of the FASB, when a seller enters into a contract with a buyer, the seller accepts certain performance obligations in exchange for the...
-
A disk 4 0 cm in diameter and 5 cm thick is to be cast of pure aluminum in an open mold casting operation. The melting temperature of aluminum = 6 6 0 \ deg C , and the pouring temperature will be 8...
-
A heat engine has a heat input of 3 Ã 104 Btu/h and a thermal efficiency of 40 percent. Calculate the power it will produce, in hp. Source 3 x 10 Btu/h 40% HE Sink
-
On January 1, 2013, Burleson Corporations projected benefit obligation was $30 million. During 2013 pension benefits paid by the trustee were $4 million. Service cost for 2013 is $12 million. Pension...
-
At January 1, 2013, NCI Industries, Inc., was indebted to First Federal Bank under a $240,000, 10% unsecured note. The note was signed January 1, 2009, and was due December 31, 2014. Annual interest...
-
On January 1, Seneca Asset Management enters into a contract with a client to provide fund management services for one year. The client is required to pay a fixed amount of $100,000 at the end of...
-
Why is the thermodynamic process also called cyclic process? Explain with suitable example.
-
A mercury manometer reads \(40 \mathrm{~cm}\) at \(30^{\circ} \mathrm{C}\) and \(1 \mathrm{~atm}\). Determine the absolute pressure, given that \(ho_{\mathrm{Hg}}=14.02 \mathrm{~g} /...
-
Define isothermal, isochoric and isobaric processes with suitable examples.

Study smarter with the SolutionInn App


