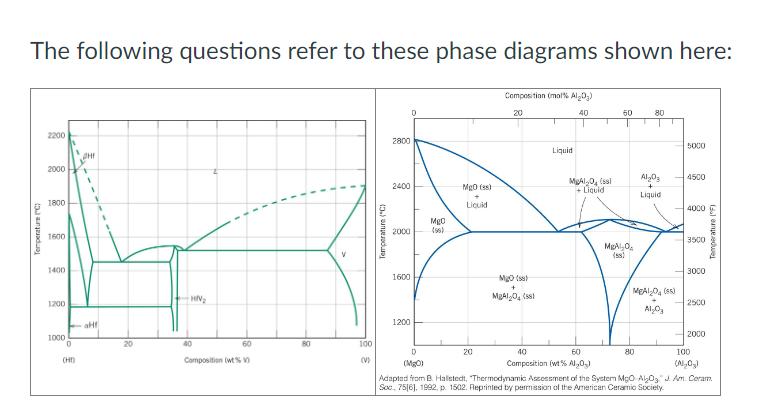
The following questions refer to these phase diagrams shown here: Temperature C 2200 2000 1800 1600...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
The following questions refer to these phase diagrams shown here: Temperature C 2200 2000 1800 1600 1400 1200 1000 0 (HD HN 40 60 Composition (wt% 0 ' T 18 ' V Temperature (°C) 2800 2400 2000 MgO (56) MgO (ss) Liquid Composition (mol% Al₂O₂) 20 1600 Mg(s) + MBAI 04 (58) 40 50 80 T 5000 Liquid MgAl-0 (ssl +Liquid Al2O3 4500 Liquid 4000 3500 MgAl (58) 3000 MEAL-O (85) 2500 Al-03 Temperature (°F) 1200 100 D 20 (V) (MgO) 40 60 Composition (wt% Al,0) 2000 80 100 Adapted from B. Halstedt, Thermodynamic Assessment of the System MgO AO Soc, 7516], 1992, p. 1502. Reprinted by permission of the American Ceramic Society (NOx) Am. Ceram. For the metallic system at 50 wt% V and 1400°C (cooled from 2000°C): A. Sketch the alloy microstructure. B. Label all phase(s) presented in your sketch. For the ceramic system: A. What is/are the intermediate compound(s) presented? B. What is/are their melting temperature(s)? Don't forget the unit. For the ceramic system: A. How many eutectic and eutectoid compositions are presented? B. For each of these compositions, where are they located in the diagram (composition & temperature)? Don't forget proper units. The following questions refer to these phase diagrams shown here: Temperature C 2200 2000 1800 1600 1400 1200 1000 0 (HD HN 40 60 Composition (wt% 0 ' T 18 ' V Temperature (°C) 2800 2400 2000 MgO (56) MgO (ss) Liquid Composition (mol% Al₂O₂) 20 1600 Mg(s) + MBAI 04 (58) 40 50 80 T 5000 Liquid MgAl-0 (ssl +Liquid Al2O3 4500 Liquid 4000 3500 MgAl (58) 3000 MEAL-O (85) 2500 Al-03 Temperature (°F) 1200 100 D 20 (V) (MgO) 40 60 Composition (wt% Al,0) 2000 80 100 Adapted from B. Halstedt, Thermodynamic Assessment of the System MgO AO Soc, 7516], 1992, p. 1502. Reprinted by permission of the American Ceramic Society (NOx) Am. Ceram. For the metallic system at 50 wt% V and 1400°C (cooled from 2000°C): A. Sketch the alloy microstructure. B. Label all phase(s) presented in your sketch. For the ceramic system: A. What is/are the intermediate compound(s) presented? B. What is/are their melting temperature(s)? Don't forget the unit. For the ceramic system: A. How many eutectic and eutectoid compositions are presented? B. For each of these compositions, where are they located in the diagram (composition & temperature)? Don't forget proper units.
Expert Answer:
Answer rating: 100% (QA)
Based on the provided image which is a magnesium oxidealuminum oxide MgOAlO pha... View the full answer

Related Book For 

Posted Date:
Students also viewed these mechanical engineering questions
-
Discuss the concepts of culture and co - culture. What is cultural centrism? Explain the benefits of cultivating cultural awareness within an organization in regard to communication and change.
-
Assume a company produces certain product. It can sell whatever it produces. Each unit is sold for 3000 AED. It can produce 80units/day. The daily estimated inputs costs are: 70 man-hours, each...
-
11. Which one is not a source for collecting references? Ans : O Proquest Ebscohost O Scopus Safe assign
-
Bright Spark Ltd is a manufacturer of electrical switchers, and uses a standard costing system standard manufacturing overhead costs per switch are based on direct labour hours and are allows:...
-
Check in each case whether the condition for the binomial approximation to the hypergeometric distribution is satisfied: (a) N = 200 and n = 12; (b) N = 500 and n = 20; (c) N = 640 and n = 30.
-
The Racial Divide The website http://vallandingham.me/racial_divide/\#pt uses data from the US Census to visualize where whites and blacks live in different cities. Figure 2.98 gives a heat map of...
-
The retained earnings account for Carlitos Inc. shows the following debits and credits. Indicate all entries required to correct the account. What is the corrected amount of retainedearnings?...
-
Performance Development Plan State the intention of moving to Customer Value Management department.
-
1. Do you think Frieds hiring methods are reasonable? Will they lead to good hires for his growing company? What are the best features of his approach (if you believe there are any)? 2. Do you think...
-
In case of IFRS adoption, why is free ridership a problem? Explain
-
At the beginning of the year, Matt's bank account had a balance of $4,589 and it stands at $4,612 today. Which one of the following statements must be FALSE when evaluat- ing the year-to-date growth...
-
Which one of the following statements is TRUE? O a . If the distribution of returns for an asset has a variance of zero, then covariance of returns between that asset and the returns any other...
-
A firm is considering the following projects. Its opportunity cost of capital is 12%. Cash Flows, $ Project Time: 0 1 2 3 4 A -5,500 +1,125 +1,125 +3,250 0 B -1,500 0 +1,500 +2,250 +3,250 C -5,500...
-
For an asset, the amount of depreciation is $800,000. The firm is in the marginal tax rate of 25 percent. Calculate the tax shield due to depreciation.
-
Which Law first legitimized bargaining for employee benefits? The Fair Labor Standards Act National Labor Relations Act Social Security Act Workers' Compensation Act
-
The following data from the just completed year are taken from the accounting records of Mason Company: Sales $ 654,000 Direct labor cost $ 80,000 Raw material purchases $ 139,000 Selling expenses $...
-
During 2012, Cheng Book Store paid $483,000 for land and built a store in Georgetown. Prior to construction, the city of Georgetown charged Cheng $1,300 for a building permit, which Cheng paid. Cheng...
-
Which of the following alkenes would yield the same alcohol from either oxymercuration-reduction or hydroboration--oxidation, and which would give different alcohols? Explain. (a) cis-2-butene (b)...
-
What aspect of alkene structure cannot be determined by ozonolysis?
-
Account for each of the following facts with an explanation. 1, 3-Cyclopentadiene is a considerably stronger carbon acid than 1, 4-pentadiene even though the acidic hydrogens in both cases are doubly...
-
A light ray travels from medium l to medium 3 as shown. For these media, A. nz > n 13 B. n3 = n nz C. n3
-
A Jens produces a sharply focused, inverted image on a screen. What will you see on the screen if the lens is removed? A. The image will be inverted and blurry. B. The image will be upright and...
-
An object and lens are positioned to form a well-focused, inverted image on a viewing screen. Then a piece of cardboard is lowered just in front of the lens to cover the top half of the lens. Using...

Study smarter with the SolutionInn App


