The ideal gas law tends to become inaccurate when Multiple Choice O the pressure is lowered...
Fantastic news! We've Found the answer you've been seeking!
Question:
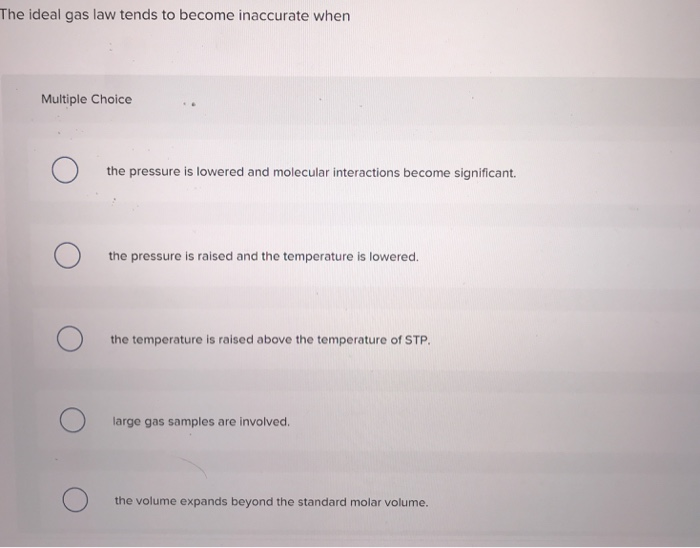
Transcribed Image Text:
The ideal gas law tends to become inaccurate when Multiple Choice O the pressure is lowered and molecular interactions become significant. the pressure is raised and the temperature is lowered. the temperature is raised above the temperature of STP. large gas samples are involved. the volume expands beyond the standard molar volume. The ideal gas law tends to become inaccurate when Multiple Choice O the pressure is lowered and molecular interactions become significant. the pressure is raised and the temperature is lowered. the temperature is raised above the temperature of STP. large gas samples are involved. the volume expands beyond the standard molar volume.
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below the temperature is ra... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
We state that the ideal gas law tends to hold best at low pressures and high temperatures. Show how the van der Waals equation simplifies to the ideal gas law under these conditions.
-
The ideal gas law relates the pressure P, volime V, and temperature T of an ideal gas: PV= nRT where " is the number of moles and R = 8.3145 J/(K. mol). Plots of pressure versus volume at constant...
-
The ideal gas law relates four variables. An empirical gas law relates two variables, assuming the other two are constant. How many empirical gas laws can be obtained? Give statements of each.
-
A 15 mm 15 mm silicon chip is mounted such that the edges are flush in a substrate. The chip dissipates 1.4 W of power uniformly, while air at 20C (1 atm) with a velocity of 25 m/s is used to cool...
-
In 1985, Japan Airlines (JAL) bought $3 billion of foreign exchange contracts at 180/$1 over 11 years to hedge its purchases of U.S. aircraft. By 1994, with the yen at about 100/$1, JAL had incurred...
-
Following the companion website access instructions above, read the "Sarbanes-Oxley Act" PowerPoint presentation, and write a one-page summary of it.
-
Redesign the fractionator of Example 6.8 for a reflux ratio that is twice the minimum. Determine the diameter of the tower, the height of packing in the stripping and rectifying sections, and the...
-
Audio House, Inc., is developing its annual financial statements at December 31, 2013. The statements are complete except for the statement of cash flows. The completed comparative balance sheets and...
-
Briefly describe TWO (2) drawbacks of NoSQL in comparison with RDBMS ?
-
Daniel B. Butler and Freida C. Butler, husband and wife, file a joint return. The Butlers live at 625 Oak Street in Corbin, KY 40701. Dan's Social Security number is 111-11-1112, and Freida's is...
-
A boat is circling a ship that is using a tracking radar. The system as shown in FIGURE (i) is given as a unity feedback system in FIGURE (ii) with a forward transfer function of the plant, G(s) =...
-
Iramba Sea Foods Investment is negotiating a Loan from Tanzania Investments Bank which has kindly requested the Company to prepare and submit their cash budget for the first half year. The following...
-
Dilemmas As a purchasing manager, you may be faced with making product decisions that conflict with your own strongly-held values and beliefs. For example: You are concerned about the environment,...
-
What is the relationship between communication and health inequality? Specify the pathways through which communication influences health inequality.
-
2. The Quickie Mart receives a shipment of batteries on Jan 20th. 15 cases are delivered. The price is $45 per case with discounts of 6% and 5% and payment terms of 1/15, n/30. a. What is the last...
-
Sarah Green has been an accountant for 12 years. She worked in a small medical practice for two years after earning her BS in Accounting and then chose to work freelance for five years. For the past...
-
and and B = ras Let A = digits of your ID. Verify each of the following (a) (AB)- = B-A-1 (c) (A)- = (A-) (e) (3A) = 3AT a, where a, a2, 93, 94, 95, 96, 97, and ag are the last eight the id that you...
-
Ball bearings are widely used in industrial applications. You work for an industrial food machinery manufacturer and your role is to design the driveshaft assembly on a new type of equipment that...
-
A cross section of a river with measurements of its depth at intervals of 40 ft: is shown in the figure. Use numerical integration to estimate the cross-sectional area of the river. 607179 49442794 62
-
The windchill temperature Twc is the perceived air temperature felt by the body on exposed skin due to the flow of air. For temperatures below 50F and wind speed higher than 3 mph, it is calculated...
-
Show that = 4/3 Do this by first creating a vector x that has the elements 2.0, 1.5, 1.1, 1.01, 1.001, 1.00001, and 1.0000001. Then, create a new vector yin which each element is determined from the...
-
Will transferable or inherent comparative advantages be more impacted by the law of one price? Why?
-
When marginal cost equals the minimum point of average variable cost, what is true about the average productivity and marginal productivity of workers?
-
What determines the distance between the average total cost and the average variable cost?

Study smarter with the SolutionInn App


