Question: The Lead-Tin phase diagram: a) Show the eutectic three phase reaction on this phase diagram. b) A Pb-45% Sn alloy is cooled from 300C
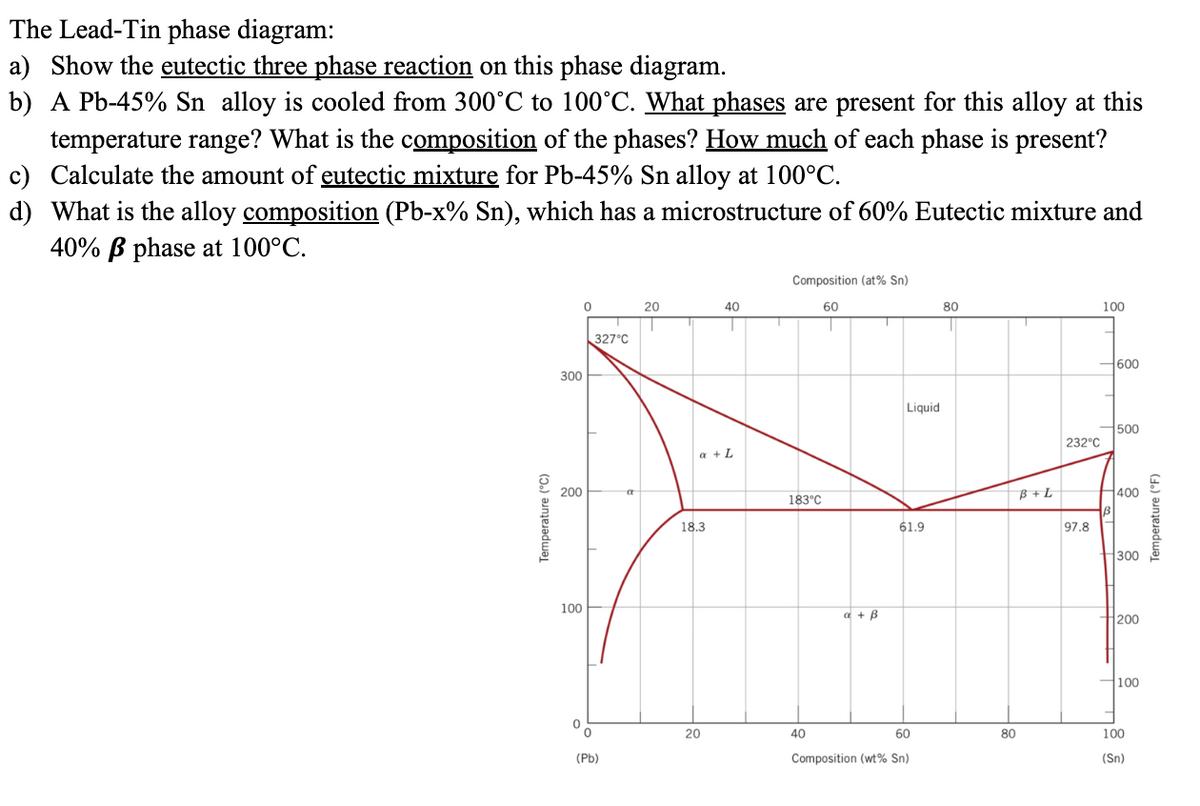
The Lead-Tin phase diagram: a) Show the eutectic three phase reaction on this phase diagram. b) A Pb-45% Sn alloy is cooled from 300C to 100C. What phases are present for this alloy at this temperature range? What is the composition of the phases? How much of each phase is present? c) Calculate the amount of eutectic mixture for Pb-45% Sn alloy at 100C. d) What is the alloy composition (Pb-x% Sn), which has a microstructure of 60% Eutectic mixture and 40% phase at 100C. Composition (at% Sn) 60 0 20 40 80 100 1 Temperature (C) 300 200 100 327C '0 (Pb) a a + L 18.3 20 183C a + B Liquid 61.9 40 60 Composition (wt% Sn) 80 B+L 232C 97.8 600 500 400 300 200 100 100 (Sn) Temperature (F)
Step by Step Solution
3.48 Rating (151 Votes )
There are 3 Steps involved in it
327c 300C d 1080 xL IG 183C A Liquid 2 B e D BL iX HI 183 1 ... View full answer

Get step-by-step solutions from verified subject matter experts


