Measured conditions for the bottoms from a depropanizer distillation unit in a small refinery are given below.
Question:
Measured conditions for the bottoms from a depropanizer distillation unit in a small refinery are given below. Using the data in Figure and assuming an ideal liquid solution (volume of mixing = 0), compute the liquid density in lb/ft3, lblgal, lb/bbl(42 gal), andkg/m3.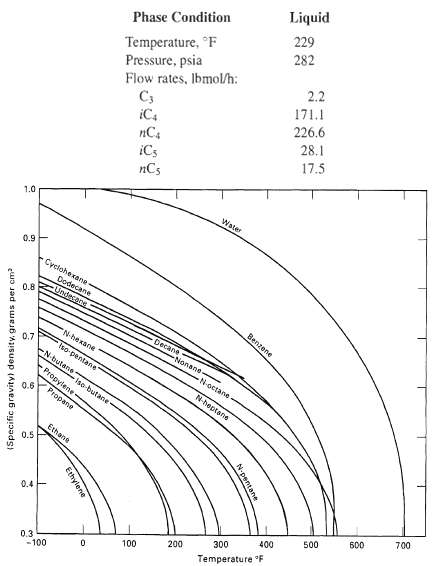
Transcribed Image Text:
Liquid 229 Phase Condition Temperature, °F Pressure, psia Flow rates, Ibmol/h: C3 iC4 nC4 iCs nCs 282 2.2 171.1 226.6 28.1 17.5 Water 1.0 0.9 Cyclohexane Dodecane Undecane 0.8 Benzene Decane N-hexane Nonane 0.7 Iso-pentane -N-butene N-octane Propylene N-heptone iso-butane Propane 0.6 Ethane 0.5 700 0.4- 600 500 400 300 200 100 Temperature F 0.3 -100 N-pentane Ethylene (Specific gravity) density, grams per cm?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
From Eq 4 Table 24 The ...View the full answer

Answered By

Naman Lakhotia
I am a master in accounts, finance and auditing. Have been helping and teaching my juniors in understanding
the basic concepts of accounting, financing and auditing.
When you understand things you learn it better and when you gulp down things you just remember it!. Once, you understand you don't forget and if you will gulp things you will forget!
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
An ideal gas expands from 1.0 m3 to 3.0 m3 at atmospheric pressure while absorbing of heat in the process. (a) The temperature of the system (1) increases, (2) stays the same, (3) decreases. Explain....
-
A rigid vessel of 0.06 m3 volume contains an ideal gas. CV = (5/2) R. at 500 K and I bar. () If heat in the amount of 15,000 J is transferred to the gas, determine its entropy change. (b) If the...
-
An ideal gas that occupies 1.2 m3 at a pressure of 1.0 105 Pa and a temperature of 27 C is compressed to a volume of 0.60 m3 and heated to a temperature of 227 C. What is the new pressure?
-
Consider the maleic anhydride process shown in Appendix B.5. Estimate the profit margin for this process using the costs of raw materials and products from Table 8.4. Figure B.5.1 Table 8.4 Costs of...
-
Using the data presented in BE12-2, assume that Columbia Ltd. intended to hold the investment until it matures. Prepare the journal entries to record (a) The purchase of the bonds on January 1, (b)...
-
Crossfade Co. issued 15-year bonds two years ago at a coupon rate of 6.9 percent. The bonds make semiannual payments. If these bonds currently sell for 94 percent of par value, what is the YTM?
-
Using the AISC equations, check if a column having the cross section shown can support an axial force of \(1500 \mathrm{kN}\). The column has a length of \(4 \mathrm{~m}\), is made from A992 steel,...
-
1. List and briefly describe the six different types of fraud symptoms. 2. Describe the different fraud symptoms present at MHZ, Inc. 3. Assume that Hal has access to the companys income statement...
-
1. Given: The sign for the Inn of the Prancing Pony in Bree-yes, it comes in pints-is fixed on the end of a beam of length 5L. If the sigh deflects too much then Gandalf will hit his head when he...
-
Spencer Duck (SSN 000-22-1111) is single and his eight-year-old son, Mitch, lives with him nine months of the year in a rented condominium at 321 Hickory Drive in Ames, Iowa. Mitch lives with his...
-
Toluene can be hydrodealkylated to benzene, but the conversion per pass through the reactor is only about 70%. Consequently, the toluene must be recovered and recycled. Typical conditions for the...
-
Isopropanol, containing 13 wt% water, can be dehydrated to obtain almost pure isopropanol at a 90% recovery by azeotropic distillation with benzene. When condensed, the overhead vapor from the column...
-
Demello & Associates records adjusting entries on an annual basis. The company has the following information available on accruals that must be recorded for the year ended December 31, 2024: 1....
-
What are the two axioms that tell you which account to debit and which account to credit?
-
What is Girsanovs theorem?
-
Explain what the stack and roll hedging strategy involves.
-
How do (a) forward and (b) futures contracts trade?
-
Explain how basis risk arises in hedging.
-
Numerically calculate a histogram of the distribution of eigenvalues of \(2 \times 2\) GOE random matrices, and show that the distribution \(P(\lambda)\) agrees with the result in Problem 5.11(e).
-
Suppose you need to answer any four of seven essay questions on a history test and you can answer them in any order. a. How many different question combinations are possible? b. What is the...
-
Suppose that the standard test statistic z = (X- 0 )/(/n) takes the value z = 2.5 and that the sample size is n = 100. How close to 0 does a value of have to be for the value of the normal...
-
What are the three contributing factors to the vapor pressure drop across a tray?
-
Fuel cell automotive systems are being considered that will require hydrogen of 95% purity. A refinery stream of 800,000 scfm (at 32 F, 1 atm), containing 72.5% H 2 , 25% CH 4 , and 2.5% C 2 H 6 ,...
-
What is meant by turndown ratio? What type of tray has the best turndown ratio? Which tray the worst?
-
Compare and contrast different deadlock prevention techniques such as resource ordering, the "hold and wait" condition, and preemptive resource allocation. Which methods are most effective for...
-
The following information is taken from Aden Company's records: Product Group Units Cost/Unit Market/Unit A 1 700 $1.10 $0.90 B 1 250 1.50 1.55 C 2 150 4.90 5.15 D 2 100 6.50 6.40 E 3 80 25.00 24.60...
-
American Food Services, Incorporated leased a packaging machine from Barton and Barton Corporation. Barton and Barton completed construction of the machine on January 1, 2024. The lease agreement for...

Study smarter with the SolutionInn App


