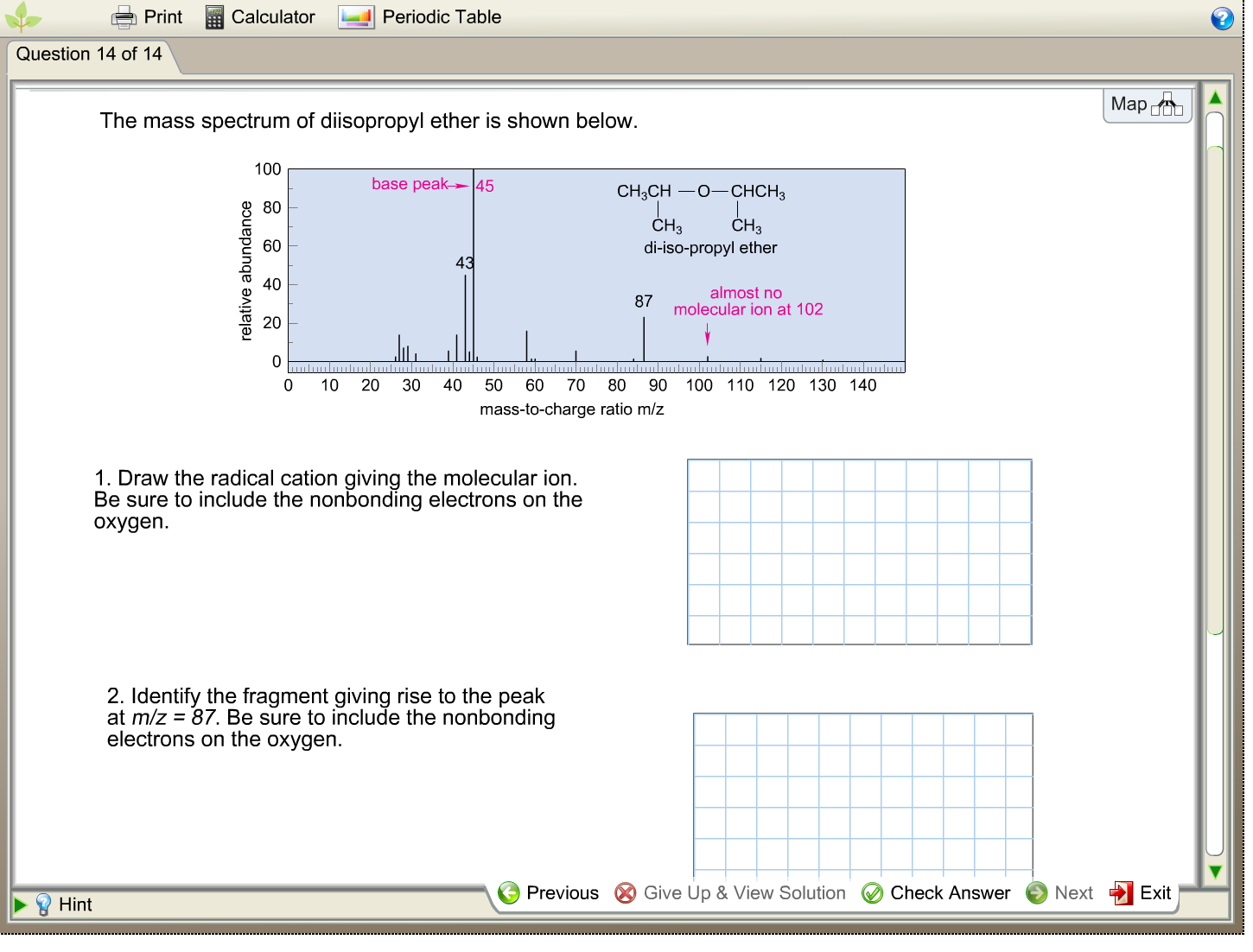
Print Question 14 of 14 Hint Calculator The mass spectrum of diisopropyl ether is shown below....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Print Question 14 of 14 Hint Calculator The mass spectrum of diisopropyl ether is shown below. relative abundance 100 80 Periodic Table 60 base peak 45 43 0 10 20 30 40 50 60 70 mass-to-charge 1. Draw the radical cation giving the molecular ion. Be sure to include the nonbonding electrons on the oxygen. 2. Identify the fragment giving rise to the peak at m/z = 87. Be sure to include the nonbonding electrons on the oxygen. Previous CH3CHO-CHCH3 CH3 CH3 di-iso-propyl ether 87 almost no molecular ion at 102 80 90 100 110 120 130 140 ratio m/z Give Up & View Solution Check Answer Next Map DOU Exit Print Question 14 of 14 Hint Calculator The mass spectrum of diisopropyl ether is shown below. relative abundance 100 80 Periodic Table 60 base peak 45 43 0 10 20 30 40 50 60 70 mass-to-charge 1. Draw the radical cation giving the molecular ion. Be sure to include the nonbonding electrons on the oxygen. 2. Identify the fragment giving rise to the peak at m/z = 87. Be sure to include the nonbonding electrons on the oxygen. Previous CH3CHO-CHCH3 CH3 CH3 di-iso-propyl ether 87 almost no molecular ion at 102 80 90 100 110 120 130 140 ratio m/z Give Up & View Solution Check Answer Next Map DOU Exit
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The mass spectrum of methyl isobutyl ether does not show a peak due to inductive cleavage, in contrast to the mass spectrum of di-sec-butyl ether (Eq. 12.31). Use what you know about carbocation...
-
The mass spectrum of unknown compound A shows a molecular ion at m/z 116 and prominent peaks at m/z 87 and m/z 101. Its UV spectrum shows no maximum above 200 nm. The IR and NMR spectra of A follow....
-
The mass spectrum of tert-butylamine follows. Use a diagram to show the cleavage that accounts for the base peak. Suggest why no molecular ion is visible in this spectrum. 100 58 80 (CHCNH2 2 40 10...
-
If the end of the cord is pulled downward with speed v C , determine the angular velocities of pulleys A and B and the speed of block D. Assume that the cord does not slip on the pulleys. Given: VC =...
-
Explain the importance of the excess-capacity issue in setting a competitive bid price.
-
Discuss the types of operational conflicts that could occur in an international context because of differences in attitudes towards time, change, material factors, and individualism. Give examples...
-
Bats are capable of navigating using the earth's field-a plus for an animal that may fly great distances from its roost at night. If, while sleeping during the day, bats are exposed to a field of a...
-
The Anson Manufacturing Company reviewed its year-end inventory and found the following items. Indicate which items should be included in the inventory balance at December 31, 2011. Give your reasons...
-
KRJ Enterprises reported a current ratio of 1.5 last year and 2.3 this year and reported a quick ratio of 1.1 last year and 1.2 this year. At the same time its days' sales in receivables remained...
-
Estimate damages for lost operation profit for years 3 through 5 from lost sales due to infringement as of June 30th, year 6, which is the estimated date of the completion of the patent infringement...
-
Baker v. Carr (1962) saw the Supreme Court enter a "political thicket" that it had been careful to avoid in the past. However, the politics of the decision extend beyond the fact that it dealt with...
-
A tuba may be treated like a tube closed at one end. If a tuba has a fundamental frequency of 45.4 Hz, determine the first three overtones. Use 343 m/s as the speed of sound in air. first overtone...
-
Determine the vector equation of the plane that contains the following two lines L1: r = (2, 3,-5) + t(5, 1, 3), ter L2:r = (2, 3, -5)+ s(0, 2,-3), SER
-
Three point charges are located on a circular arc as shown in the figure below. (Take r 4.28 cm. Let to the right be the +x direction and up along the screen be the +y direction.) = +3.00 nC C -2.00...
-
A proton with a kinetic energy of 1.80 keV, that is at height 21.6 cm above a horizontal charged nonconducting plate with surface charge density 3.98 C/m2, is fired horizontally across this plate....
-
Given three capacitors, C = 2.0 F, C = 1.5 F, and C3 = 3.0 F. What arrangement of parallel and series connections with a 28-V battery will give the minimum voltage drop across the 2.0 F capacitor?...
-
What are Financial Statements of a company and what do they tell about a company?
-
The executor of Gina Purcells estate has recorded the following information: Assets discovered at death (at fair value): Cash . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
A graduate student was following a procedure to make 3-propylcyclohexa-1,4-diene. During the workup procedure, his research adviser called him into her office. By the time the student returned to his...
-
(a) When n-heptane burns in a gasoline engine, the combustion process takes place too quickly. The explosive detonation makes a noise called knocking. When 2,2,4-trimethylpentane (isooctane) is...
-
Show how you would accomplish each of the following synthetic conversions. (a) trans-but-2-ene ¡ trans-1,2-dimethylcyclopropane (b) (c) cyclopentene> Br CI cyclohexanol> CI
-
Salen Company finances some of its current operations by assigning accounts receivable to a finance company. On July 1, 2015, it assigned, under guarantee, specific accounts amounting to 150,000,000....
-
Presented below is information related to the Accounts Receivable accounts of Gulistan Inc. during the current year 2015. 1. An aging schedule of the accounts receivable as of December 31, 2015, is...
-
Bill Jovi is reviewing the cash accounting for Nottleman, Inc., a local mailing service. Jovis review will focus on the petty cash account and the bank reconciliation for the month ended May 31,...

Study smarter with the SolutionInn App


