The melting point of fluorine, F2, is-220 C and the melting point of chlorine, Cl, is...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
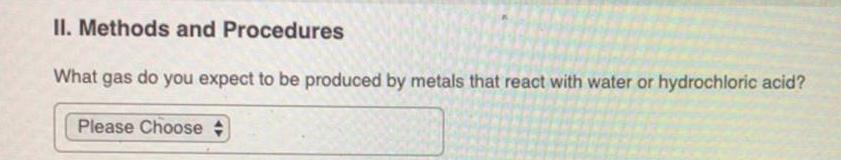
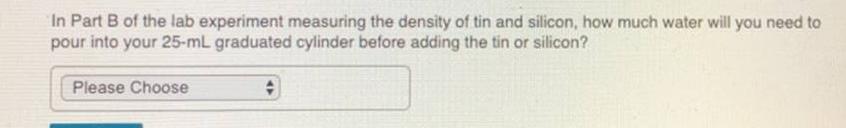
The melting point of fluorine, F2, is-220 °C and the melting point of chlorine, Cl₂, is -103 °C. Will the melting point of bromine, Brą, be higher or lower than that of chlorine? O lower Ohigher II. Methods and Procedures What gas do you expect to be produced by metals that react with water or hydrochloric acid? Please Choose What test will you use to confirm the presence of this gas? Please Choose Submit Request Answer # In Part B of the lab experiment measuring the density of tin and silicon, how much water will you need to pour into your 25-mL graduated cylinder before adding the tin or silicon? Please Choose # The melting point of fluorine, F2, is-220 °C and the melting point of chlorine, Cl₂, is -103 °C. Will the melting point of bromine, Brą, be higher or lower than that of chlorine? O lower Ohigher II. Methods and Procedures What gas do you expect to be produced by metals that react with water or hydrochloric acid? Please Choose What test will you use to confirm the presence of this gas? Please Choose Submit Request Answer # In Part B of the lab experiment measuring the density of tin and silicon, how much water will you need to pour into your 25-mL graduated cylinder before adding the tin or silicon? Please Choose #
Expert Answer:
Answer rating: 100% (QA)
Answer Part B The melting point of bromine Br2 will be higher than that of chlorine Cl2 Part C Metals that react with water or hydrochloric acid are e... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Will the banks ROA next year be higher or lower if market interest rates are higher? (Use the T-bill rate as a proxy for market interest rates.) Why? The information provided did not assume any...
-
Do you expect to see larger solidification shrinkage for a material with a bcc crystal structure or fee? Explain.
-
Is the p50 higher or lower than normal in (a) Hemoglobin Yakima and (b) Hemoglobin Kansas? Explain.
-
Find the coordinates of the vertices and the foci of the given hyperbolas. Sketch each curve. 9y 2 16x 2 = 9
-
Your company has decided to switch from its computerized transaction-based accounting system to an ERP system. Your boss understands that this system is capable of tracking accounting and other...
-
Which approaches to interpersonal communication are likely to be most useful at Trader Joes?
-
The flowrate in the clay-lined channel \((n=0.025)\) shown in Fig. P10.54 is to be \(300 \mathrm{ft}^{3} / \mathrm{s}\). To prevent erosion of the sides, the velocity must not exceed \(5 \mathrm{ft}...
-
A refrigerator used by a meat processor has a cost of $86,750, an estimated residual value of $7,500, and an estimated useful life of 25 years. What is the amount of the annual depreciation computed...
-
Research the civil case Katz, et al. v. Panera Bread Co. and answer the following questions: Give a brief description of the lawsuit. Did Panera properly display the contents of the drink? Explain...
-
Compare and contrast the leadership styles exhibited by Steve Jobs and Tim Cook.
-
Ron is repaying a loan with payments of 1 at the end of each year for n years. The annual effective interest rate on the loan is i. The amount of interest paid in year t plus the amount of principal...
-
True Or False The defendant is liable for the full extent of the plaintiffs harm even if they could not have reasonably anticipated that harm.
-
Opening statements a. allow an attorney to introduce the parties. b. are not particularly important. c. are considered part of the evidence. d. all of the above.
-
Why is it so difficult to hold a defendant liable for the infliction of emotional distress?
-
The doctrine of transferred intent is applicable to a. battery. b. assault. c. false imprisonment. d. all of the above.
-
If a person is trapped by another, but has one way out, is that considered false imprisonment?
-
4. You are saving for retirement. Suppose you are comfortable with an annual salary of $65,000 per year. You'd like to retire in 47 years and then plan to live for another 25 years. (a) What would be...
-
Bonus shares can be issued out of revenue reserves. True/False?
-
A compressed-gas cylinder contains 1.00 10 3 g of argon gas. The pressure inside the cylinder is 2050. psi (pounds per square inch) at a temperature of 18 o C. How much gas remains in the cylinder...
-
The following illustration displays the relative number of species when an acid, HA, is added to water. a. Is HA a weak or strong acid? How can you tell? b. Using the relative numbers given in the...
-
In the production of printed circuit boards for the elec¬tronics industry, a 0.60-mm layer of copper is laminated onto an insulating plastic board. Next, a circuit pattern made of a chemically...
-
Is the word anxiety a candidate for creating a stable pattern? If so, give reasons.
-
Define the real meaning of anxiety. What are the different meanings of this word?
-
Can you list six important benefits of this pattern?

Study smarter with the SolutionInn App


