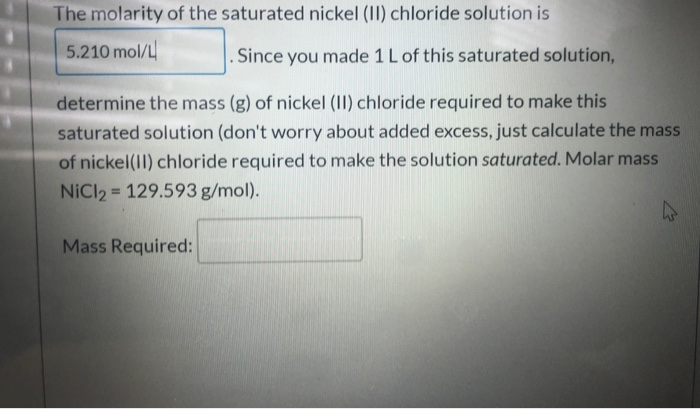
* The molarity of the saturated nickel (II) chloride solution is 5.210 mol/L . Since you...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
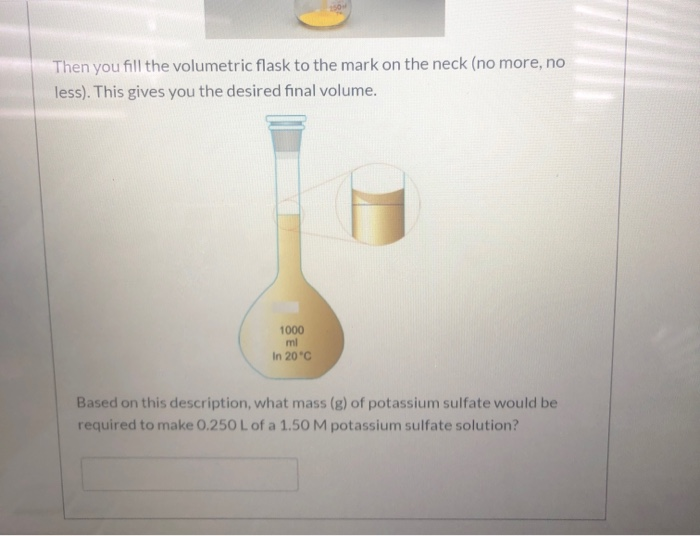
* The molarity of the saturated nickel (II) chloride solution is 5.210 mol/L . Since you made 1 L of this saturated solution, determine the mass (g) of nickel (II) chloride required to make this saturated solution (don't worry about added excess, just calculate the mass of nickel(II) chloride required to make the solution saturated. Molar mass NiCl₂ = 129.593 g/mol). Mass Required: 4 When preparing a solution of known concentration (stock solution) from the ionic solid, we must calculate the mass solid required to make the volume and concentration of solution desired. For example, if I wanted to make 1.5 L of a 0.50 M solution of sodium iodide (Nal), I would first find the number of moles of Nal required for the solution: 1.5 L (0.50 mol/L) = 0.75 mol Nal Next, I would use the molar mass of Nal to determine the mass of Nal required: 0.75 mol Nal (149.89 g/mol) = 112.4 g Nal (not worrying about sig figs) (PICTURES BELOW DO NOT REPRESENT THE SUBSTANCE OR QUANTITIES IN THE EXAMPLE). To prepare this solution, we would measure out the Nal using an analytical balance. We would carefully transfer this mass of Nal (not pictured) to a volumetric flask: FUNDAMENTAL Then you fill the volumetric flask to the mark on the neck (no more, no less). This gives you the desired final volume. 1000 ml In 20°C Based on this description, what mass (g) of potassium sulfate would be required to make 0.250 L of a 1.50 M potassium sulfate solution? * The molarity of the saturated nickel (II) chloride solution is 5.210 mol/L . Since you made 1 L of this saturated solution, determine the mass (g) of nickel (II) chloride required to make this saturated solution (don't worry about added excess, just calculate the mass of nickel(II) chloride required to make the solution saturated. Molar mass NiCl₂ = 129.593 g/mol). Mass Required: 4 When preparing a solution of known concentration (stock solution) from the ionic solid, we must calculate the mass solid required to make the volume and concentration of solution desired. For example, if I wanted to make 1.5 L of a 0.50 M solution of sodium iodide (Nal), I would first find the number of moles of Nal required for the solution: 1.5 L (0.50 mol/L) = 0.75 mol Nal Next, I would use the molar mass of Nal to determine the mass of Nal required: 0.75 mol Nal (149.89 g/mol) = 112.4 g Nal (not worrying about sig figs) (PICTURES BELOW DO NOT REPRESENT THE SUBSTANCE OR QUANTITIES IN THE EXAMPLE). To prepare this solution, we would measure out the Nal using an analytical balance. We would carefully transfer this mass of Nal (not pictured) to a volumetric flask: FUNDAMENTAL Then you fill the volumetric flask to the mark on the neck (no more, no less). This gives you the desired final volume. 1000 ml In 20°C Based on this description, what mass (g) of potassium sulfate would be required to make 0.250 L of a 1.50 M potassium sulfate solution?
Expert Answer:

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemistry questions
-
Intentionally underestimating revenue when preparing a budget is known as a. Low-balling. b. Forecasting. c. Projecting. d. Predicting.
-
Calculate the ionic strength of a solution that is 0.040 mol kg-I in K3 [Fe (CN) 6J (aq), 0.030 mol kg-1 in KCI (aq), and 0.050 mol kg3 in NaBr (aq).
-
When methyl iodide at 0.I M concentration is allowed to react with sodium ethoxide at 0.1 M concentration in ethanol solution, the product ethyl methyl ether is obtained in good yield. Explain why...
-
What Codification citation indicates how unrealized gains or losses from changes in fair value affect net income when investments are classified as available-for-sale?
-
Spice asks Meyers to quantify price changes from changes in interest rates. To illustrate, Meyers computes the value change for the fixed-rate note in the table. Specifically, he assumes an increase...
-
What it R&D, and what is its importance to marketers and the product development process?
-
Following is a TI-84 Plus display showing a 95% confidence interval for 1. a. What is the slope of the least-squares regression line? b. How many degrees of freedom are there? c. How many points are...
-
King Peak Company produces one security door model. A partially complete table of its costs follows: Required: 1. Complete the table. 2. King Peak sells its doors for $200 each. Prepare a...
-
2 pts In 2022, DCP Corporation had the following transactions: Sold building with a book value of $760,000 for $820,000 . Purchased machinery for $200,000 Purchased investments for $640,000 What is...
-
On December 31, 2023, Stilton Service Companys year-end, the unadjusted trial balance included the following items: Required 1. Prepare the adjusting entry on the books of Stilton Service Company to...
-
22 1 point Which atom has the smallest number of neutrons C Oxygen-16 O Fluorine-19 O Neon-20 C Nitrogen-14 O Carbon-14 Previous sa Next
-
Use a simple diagram of the loanable funds market to show how a sudden fall in income of the consumers or households of a country (with no other changes taking place in the economy) affect the real...
-
Rewrite the if logic in the code below as one nested if statement. int x = scan.nextInt(); int y; if (x == 10) y = 5; if (x == 20) y = 6; if (x!= 10 && x != 20) y = 7;
-
A beam AB of span 1 2 m is simply supported at its ends is subjected to a point load of 4 0 kN at C at a distance of 4 m from left end. Using moment area method, Compute thedeflection at the point C...
-
It might be a good idea to buy a mobile home to use while you are going to school. Suppose you live in the mobile home for four years, but your roommate also pays you $3,000 a year, paid to you at...
-
Frank owns a $ 5 0 0 , 0 0 0 life insurance policy on his wife Tonya. Frank's son, Sam, is designated as the beneficiary of the policy. Within 9 M of Tonya's death, Sam disclaims his interest in the...
-
Problem 2: [20 pts] (Floyd-Warshall algorithm) Let G = (V, E) be the directed weighted graph shown below with its asso- ciated weighted adjacency matrix. Solve the all-pairs shortest-path problem on...
-
Suppose that you are part of a virtual team and must persuade other team members on an important matter (such as switching suppliers or altering the project deadline). Assuming that you cannot visit...
-
Diane is a single taxpayer who qualifies for the earned income credit. Diane has two qualifying children who are 3 and 5 years old. During 2012, Diane's wages are $17,100 and she receives dividend...
-
Professor Patricia (Patty) Pate is retired from the PalmSprings Culinary Arts Academy (PSCAA). She is a single taxpayer and is 68 years old. Patty lives at 98 Colander Street, Henderson, NV 89052....
-
Brian and Kim have a 12-year-old child, Stan. For 2012, Brian and Kim have taxable income of $52,000, and Stan has nonqualifying dividend income of $4,500 and investment expenses of $250. No election...
-
The presence of air in the brake system fluid causes the brake pedal to feel __________________.
-
Three main components of pst brake antiskid systems are __________________, __________________ and a __________________.
-
__________________ pressure bleeding is not an option for power brakes; the air trapped in the brake system would be forced into the main hydraulic system, which should operate without air in the...

Study smarter with the SolutionInn App


