The molecular formula of an unknown peptide is C 20 H 34 N 6 O 6 .
Question:
The molecular formula of an unknown peptide is C20H34N6O6.
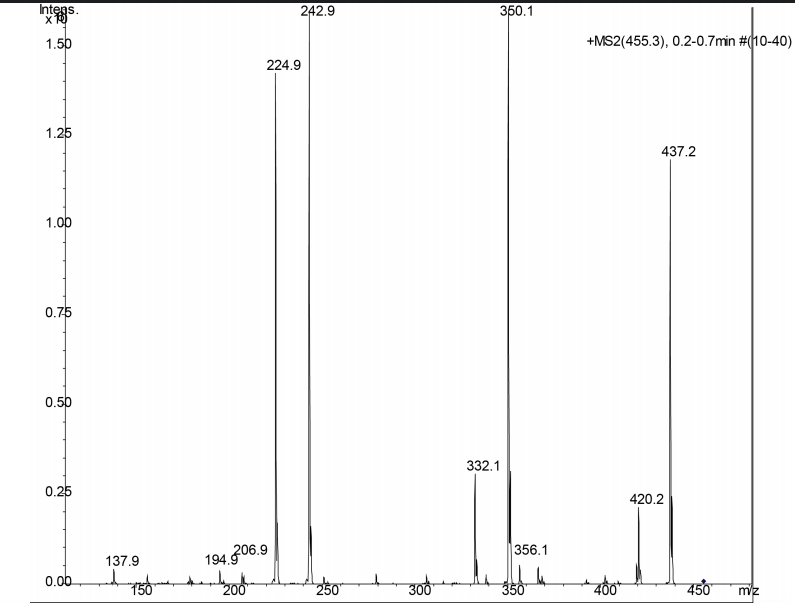
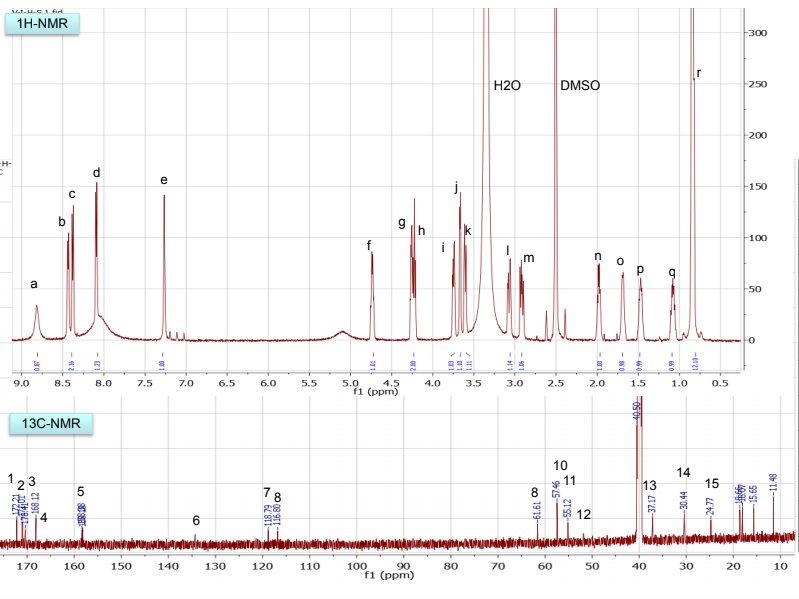
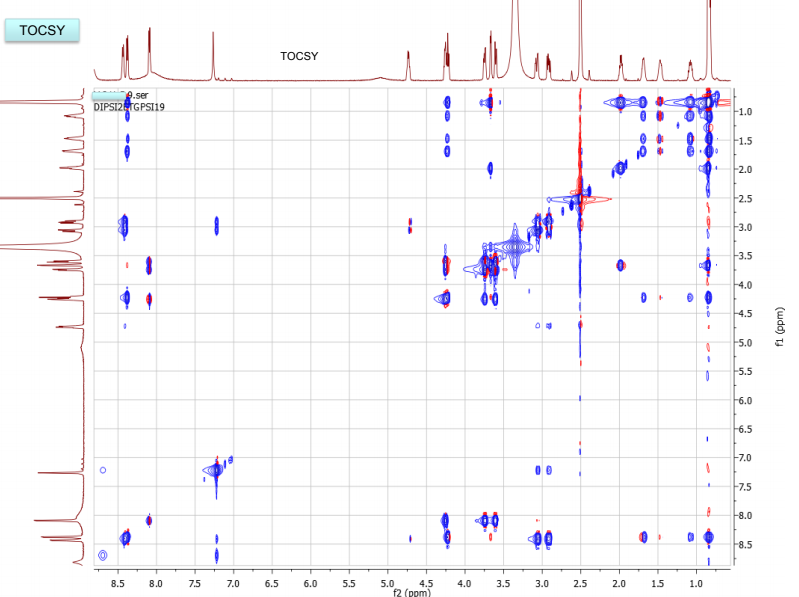
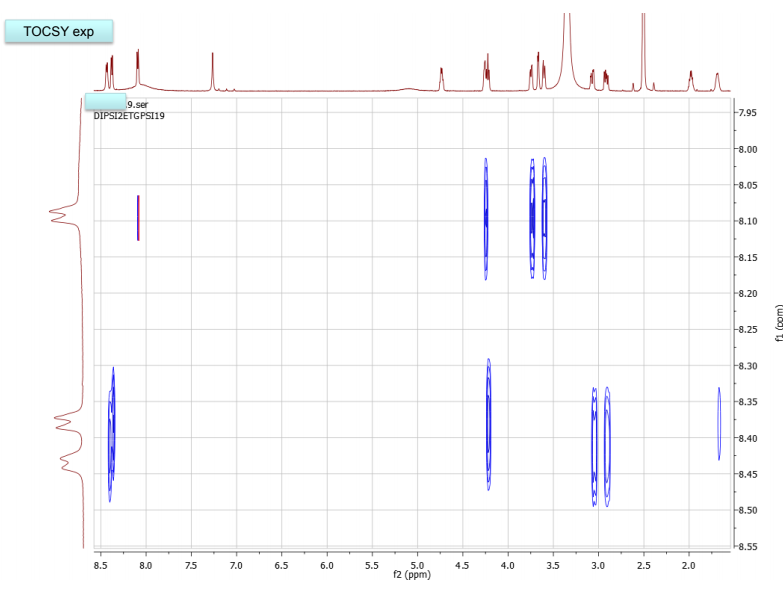
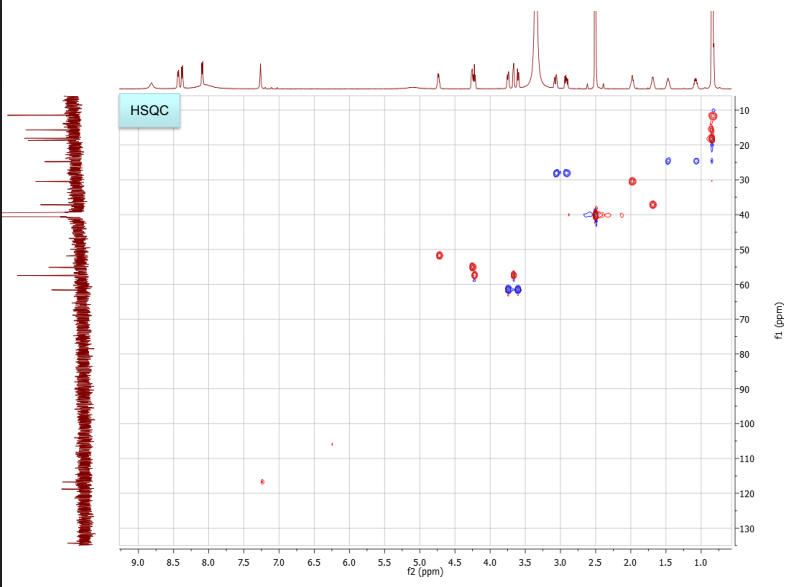
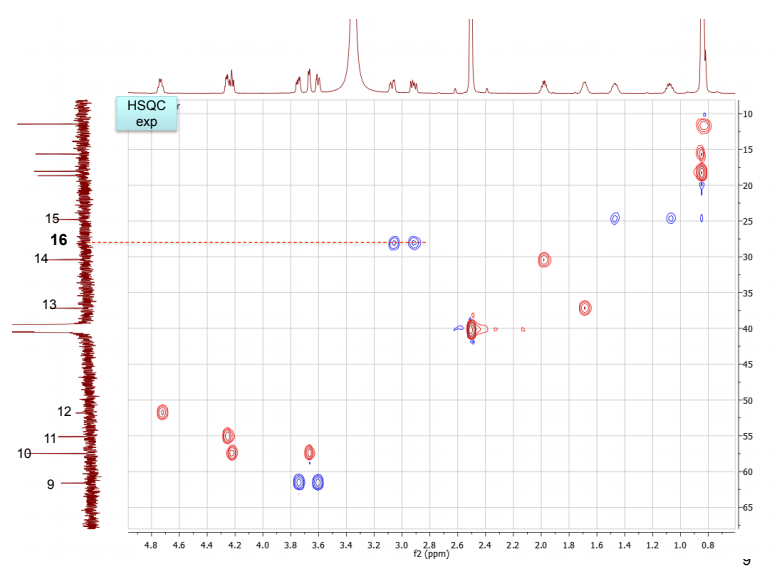
NMR spectra, taken in d6-DMSO/D2O on our 600 MHz NMR are provided. DMSO gives a proton signal at 2.49 and a carbon signal at 40 ppm. In addition to drawing and labeling the structure, answer or address the following points. The MS/MS data was acquired in (+) ESI mode and the precursor mass was 455 m/z.
a. What are the amino acids of the peptide and what is their sequence. Draw the MS/MS fragment structures that lead to the sequence. Where there any fragment ions not present? If so, draw the structures. Label each structure using proper peptide fragmentation nomenclature (as discussed in class). Make a table with structures of all a-c and x-y ions, as found in the MS.
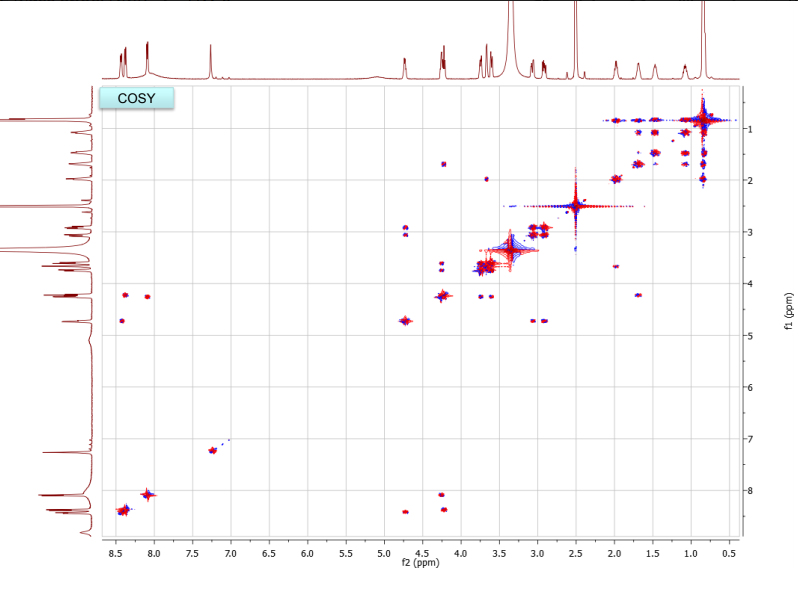
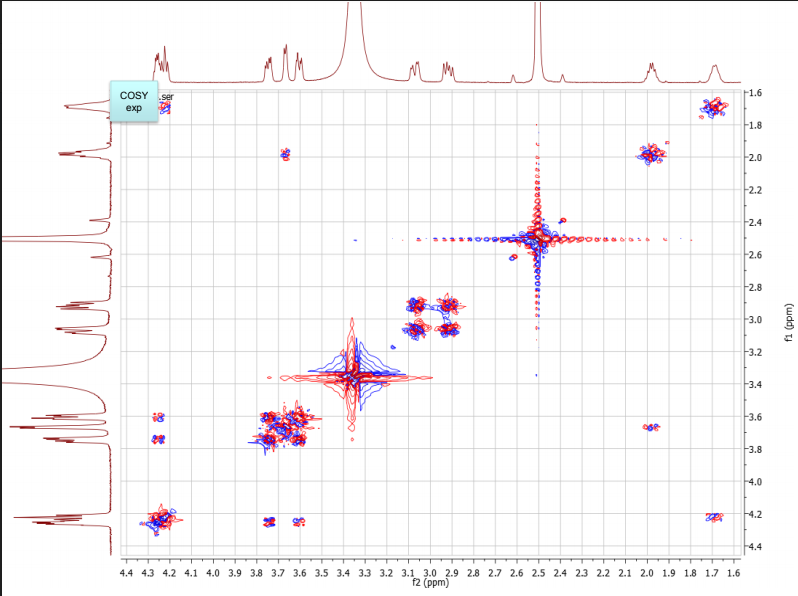
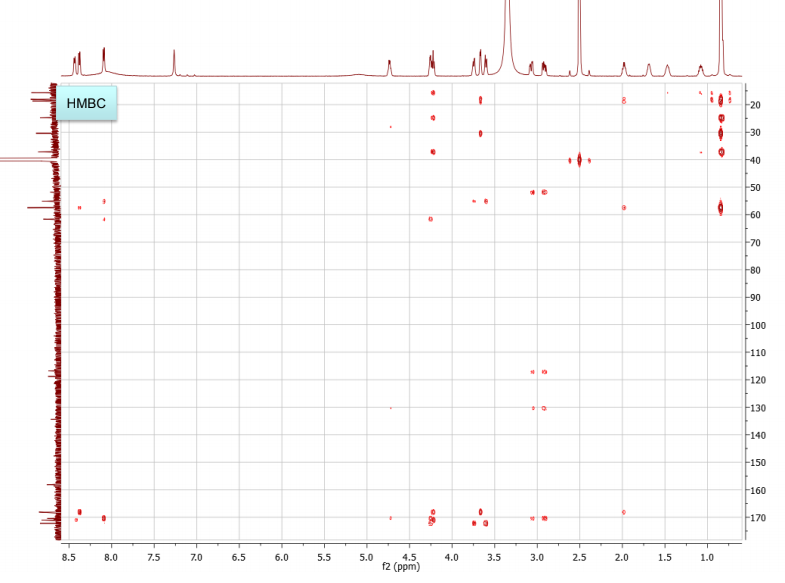
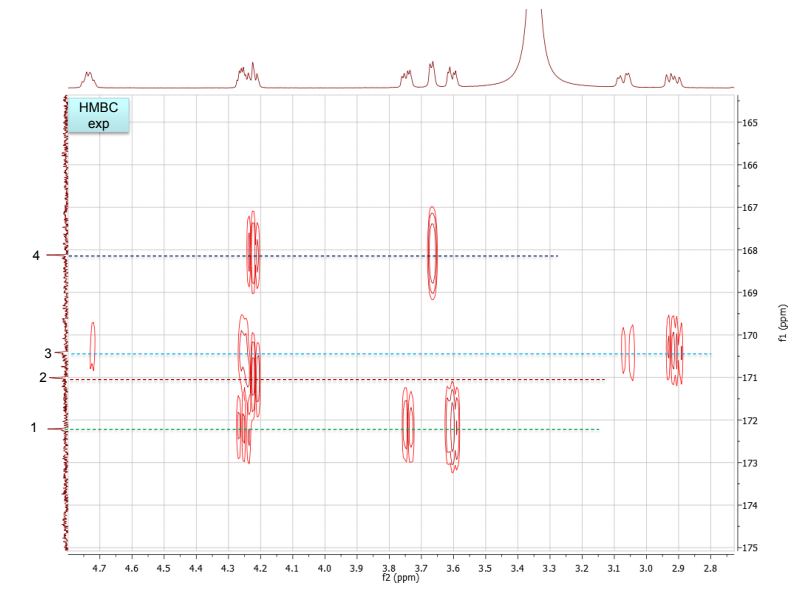
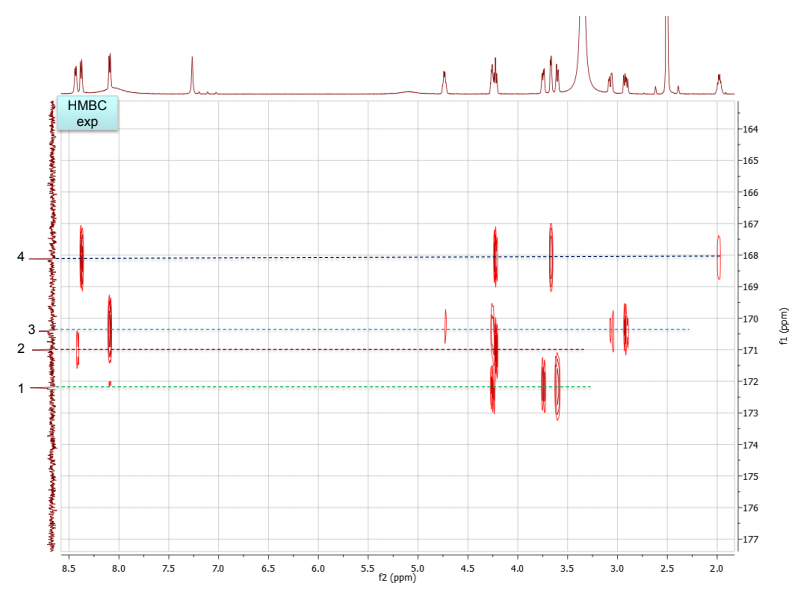
b. Draw the structure for the peptide and assign all protons (letters a-r as given on the H-NMR spectrum) and carbons 1-16. Draw your structure on the last HMBC spectrum and show all distinctive correlations (for example, bH to C=O, aH-C=O and NH to C=O) which helped assign or confirm your proposed structure.Notes:1. Carbon 16 does not show up in the 13C-NMR spectrum, but is apparent from correlations on the HSQC spectra.2. Also, as can be seen in the HSCQ spectra, C10 is actually from two overlapping carbons.3. In the HMBC there is a missing C-H correlation between 115 and 125 ppm on the carbon scale.4. One of the correlations between b protons and its corresponding carbonyl carbon is not intense enough to see on these plots.
c. If there are still any ambiguities in your assignments, describe what other experiment you might run to confirm your assignment on the bottom of your MS table or the bottom of the HMBC spectrum you submit.

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward




