The purpose of this lab is to experimentally determine the equilibrium constant, Kc. for the following chemical
Fantastic news! We've Found the answer you've been seeking!
Question:
The purpose of this lab is to experimentally determine the equilibrium constant, Kc. for the following chemical reaction:





Transcribed Image Text:
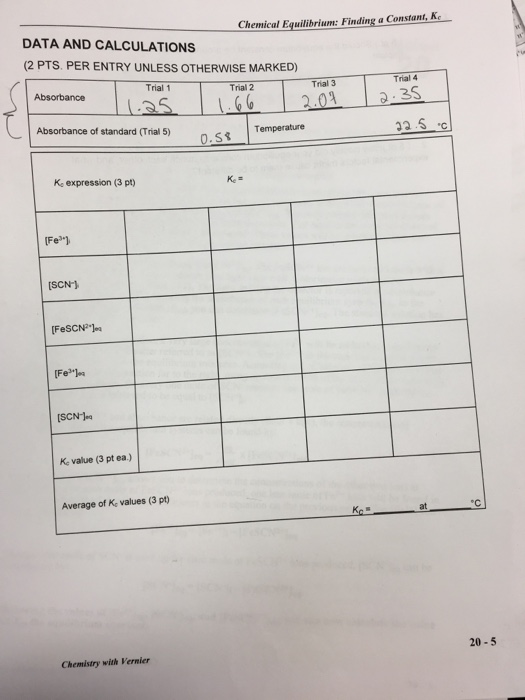
Fe³+ (aq) + SCN- (aq) →→→→ FeSCN² (aq) iron(III) thiocyanate thiocyanoiron(III) When Fe³+ and SCN are combined, equilibrium is established between these two ions and the FeSCN2+ ion. In order to calculate Ke for the reaction, it is necessary to know the concentrations of all ions at equilibrium: [FeSCN2+leq [SCN-leg, and [Fe³ leq. You will prepare four equilibrium systems containing different concentrations of these three ions. The equilibrium concentrations of the three ions will then be experimentally determined. These values will be substituted into the equilibrium constant expression to see if K, is indeed constant. You will use a Colorimeter to determine [FeSCN²+leq The FeSCN2+ ion produces solutions with a red color. Because the red solutions absorb blue light very well, Colorimeter users will be instructed to use the 470 nm (blue) LED. The light striking the detector is reported as absorbance or percent transmittance. By comparing the absorbance of each equilibrium system, Aeg, to the absorbance of a standard solution, Astd you can determine [FeSCN2+ Jeq. The standard solution has a known FeSCN²+ concentration. To prepare the standard solution, a very large concentration of Fe³+ will be added to a small initial concentration of SCN (hereafter referred to as [SCN-1;. The [Fe] in the standard solution is 100 times larger than [Fe³+] in the equilibrium mixtures. According to LeChatelier's principle, this high concentration forces the reaction far to the right, using up nearly 100% of the SCN-ions. According to the balanced equation, for every one mole of SCN reacted, one mole of FeSCN2+ is produced. Thus [FeSCN2+ Istd is assumed to be equal to [SCN-]i. Assuming [FeSCN2+] and absorbance are related directly (Beer's law), the concentration of FeSCN2+ for any of the equilibrium systems can be found by: Aeq [FeSCN²+ leq= x [FeSCN²+ ]std Astd Knowing the [FeSCN²+ Jeg allows you to determine the concentrations of the other two ions at equilibrium. For each mole of FeSCN2+ ions produced, one less mole of Fe+ ions will be found in the solution (see the 1:1 ratio of coefficients in the equation on the previous page). The [Fe³+] can be determined by: [Feleq=[Feli-[FeSCN²+ leq Because one mole of SCN is used up for each mole of FeSCN2+ ions produced, [SCN-Jeq can be determined by: [SCN-leq - [SCN-li-[FeSCN²+ leq = Knowing the values of [Feleqs [SCN-leqs and [FeSCN2+ legs you can now calculate the value of Ke the equilibrium constant. OBJECTIVE In this experiment, you will determine the equilibrium constant, Ke, for the following chemical reaction: MATERIALS Fe³+ (aq) + SCN (aq) →→→ FeSCN²+ (aq) iron(III) thiocyanate thiocyanoiron (III) Vernier LabQuest LabQuest App Vernier Colorimeter 6 plastic cuvettes five 20 x 150 mm test tubes three 100 mL beakers thermometer PROCEDURE 1. Obtain and wear goggles. 2. Label four 20 x 150 mm test tubes 1-4. Pour about 30 mL of 0.0020 M Fe(NO3)3 into a clean, dry 100 ml beaker. Pipet 5.0 mL of this solution into each of the four labeled test tubes. Use a pipet pump or bulb to pipet all solutions. CAUTION: Fe(NO)s solutions in this experiment are prepared in 1.0 M HNO, and should be handled with care. Pour about 25 mL of the 0.0020 M KSCN into another clean, dry 100 mL beaker. Pipet 2, 3, 4 and 5 mL of this solution into Test Tubes 1-4, respectively. Obtain about 25 mL of distilled water in a 100 ml beaker. Then pipet 3, 2, 1 and 0 mL of distilled water into Test Tubes 1-4, respectively, to bring the total volume of each test tube to 10 mL. Mix each solution thoroughly with a stirring rod. Be sure to clean and dry the stirring rod after each mixing. Measure and record the temperature of one of the above solutions to use as the temperature for the equilibrium constant, Ke. Volumes added to each test tube are summarized below: Test Tube Number 1 2 3 4 Fe(NO3) (mL) 20-2 5 0.0020 M KSCN 0.0020 M Fe(NO3)3 (in 1.0 M HNO3) 0.200 M Fe(NO3)3 (in 1.0 M HNO3) four pipets pipet bulb or pipet pump tissues (preferably lint-free). 5 5 5 KSCN (mL) 2 3 4 5 H₂O (mL) 3. Prepare a standard solution of FeSCN2+ by pipetting 18 mL of 0.200 M Fe(NO3)3 into a 20x150 mm test tube labeled "5". Pipet 2 mL of 0.0020 M KSCN into the same test tube. Stir thoroughly. • Wipe the outside of each cuvette with a lint-free tissue. • Handle cuvettes only by the top edge of the ribbed sides. 3 2 1 0 4. Prepare a blank by filling a cuvette 3/4 full with distilled water. To correctly use cuvettes, remember: • Dislodge any bubbles by gently tapping the cuvette on a hard surface. Always position the cuvette so the light passes through the clear sides. Chemistry with Vernier Chemical Equilibrium: Finding a Constant, Ke 5. Connect the Colorimeter to LabQuest and choose New from the File menu. 6. Calibrate the Colorimeter. a. Place the blank in the cuvette slot of the Colorimeter and close the lid. b. Press the button on the Colorimeter to select a wavelength of 470 nm. Press the CAL button on the Colorimeter. When the LED stops flashing, the calibration is complete. 7. Set up the data collection mode. a. On the Meter screen, tap Mode. Change the mode to Events with Entry. b. Enter the Name (Concentration) and Units (mol/L). Select OK. 8. You are now ready to collect absorbance data for the four equilibrium systems and the standard solution. a. With the Test Tube 1 solution, fill a new cuvette 3/4 full, and place it in the device. b. When the value displayed on the screen has stabilized, tap Keep and enter 1 (the trial number). Select OK. The absorbance and concentration values have now been saved for the first solution. Write the absorbance value on the report sheet for trial 1. c. With the Test Tube 1 solution, fill a new cuvette 3/4 full with Test Tube 2 solution, and place it in the device. Press run twice, and then "append". After the reading stabilizes, tap Keep and enter 2. Select OK to continue. Again, record the absorbance value for Trial 2, in your data table. d. Repeat the procedure to find the absorbance of the solutions in Test Tubes 3, 4, and 5 (the standard solution) with a new cuvette for each. Record the absorbance values, for each Trial, in your data table. e. Stop data collection. f. Examine the data points on the displayed graph. YOU SHOULD HAVE A STRAIGHT LINE INCREASING UP TO THE RIGHT. If you do not please contact your instructor!! g. If you want to check any data values, tap any data point. As you tap a data point, the absorbance and concentration values are displayed to the right of the graph. h. Dispose of all solutions in the waste container. Cuvettes and pipettes are also disposable. Lean any Sharpie off of the glassware.. PROCESSING THE DATA 1. Write the K expression for the reaction in the Data and Calculation table. 2. Calculate the initial concentration of Fe³+, based on the dilution that results from adding KSCN solution and water to the original 0.0020 M Fe(NO3)3 solution. See Step 2 of the procedure for the volume of each substance used in Trials 1-4. Calculate [Fe3+]; using the equation: Fe(NO3)3 mL [Feli= total mL This should be the same for all four test tubes. × (0.0020 M) LabQuest 20 3. Calculate the initial concentration of SCN", based on its dilution by Fe(NO3)3 and water: KSCN mL [SCN-li total ml × (0.0020 M) In Test Tube 1, [SCN-];-(2 mL/10 mL)(0.0020 M) = 0.00040 M. Calculate this for the other three test tubes. 4. [FeSCN2Jeq is calculated using the formula: Aeg X Astd x [FeSCN²+]std [FeSCN2 Jeq where Aeq and Asd are the absorbance values for the equilibrium and standard test tubes, respectively, and [FeSCN2 Isid (1/10)(0.0020) = 0.00020 M. Calculate [FeSCN²leq for each of the four trials. 5. [Feleq: Calculate the concentration of Fe³+ at equilibrium for Trials 1-4 using the equation: [Fe³ leq=[Fe³+]-[FeSCN²* Jeq 6. [SCN-Jeq: Calculate the concentration of SCN- at equilibrium for Trials 1-4 using the equation: [SCN-Jeq [SCN-]-[FeSCN2+ leq 7. Calculate K for Trials 1-4. Be sure to show the Ke expression and the values substituted in for each of these calculations. 8. Using your four calculated K, values, determine an average value for Ke. How constant were your Ke values? DATA AND CALCULATIONS (2 PTS. PER ENTRY UNLESS OTHERWISE MARKED) Trial 1 1.25 Absorbance of standard (Trial 5) Absorbance K. expression (3 pt) [Fe] [SCN-] [FeSCN²] [Fe] [SCN-Jea K value (3 pt ea.) Average of K, values (3 pt) Chemistry with Vernier Chemical Equilibrium: Finding a Constant, Ke Trial 2 1.66 0.58 Ke= Trial 3 2.07 Temperature Trial 4 2.35 32.5 c 76 °C 20-5 LabQuest 20 1. (5 pt.) Should the equilibrium constant be around the same for all of the test solution Explain. 2. (5 pt.)How closely did your equilibrium constants match each other, and what experimental factors might cause them to be different? Fe³+ (aq) + SCN- (aq) →→→→ FeSCN² (aq) iron(III) thiocyanate thiocyanoiron(III) When Fe³+ and SCN are combined, equilibrium is established between these two ions and the FeSCN2+ ion. In order to calculate Ke for the reaction, it is necessary to know the concentrations of all ions at equilibrium: [FeSCN2+leq [SCN-leg, and [Fe³ leq. You will prepare four equilibrium systems containing different concentrations of these three ions. The equilibrium concentrations of the three ions will then be experimentally determined. These values will be substituted into the equilibrium constant expression to see if K, is indeed constant. You will use a Colorimeter to determine [FeSCN²+leq The FeSCN2+ ion produces solutions with a red color. Because the red solutions absorb blue light very well, Colorimeter users will be instructed to use the 470 nm (blue) LED. The light striking the detector is reported as absorbance or percent transmittance. By comparing the absorbance of each equilibrium system, Aeg, to the absorbance of a standard solution, Astd you can determine [FeSCN2+ Jeq. The standard solution has a known FeSCN²+ concentration. To prepare the standard solution, a very large concentration of Fe³+ will be added to a small initial concentration of SCN (hereafter referred to as [SCN-1;. The [Fe] in the standard solution is 100 times larger than [Fe³+] in the equilibrium mixtures. According to LeChatelier's principle, this high concentration forces the reaction far to the right, using up nearly 100% of the SCN-ions. According to the balanced equation, for every one mole of SCN reacted, one mole of FeSCN2+ is produced. Thus [FeSCN2+ Istd is assumed to be equal to [SCN-]i. Assuming [FeSCN2+] and absorbance are related directly (Beer's law), the concentration of FeSCN2+ for any of the equilibrium systems can be found by: Aeq [FeSCN²+ leq= x [FeSCN²+ ]std Astd Knowing the [FeSCN²+ Jeg allows you to determine the concentrations of the other two ions at equilibrium. For each mole of FeSCN2+ ions produced, one less mole of Fe+ ions will be found in the solution (see the 1:1 ratio of coefficients in the equation on the previous page). The [Fe³+] can be determined by: [Feleq=[Feli-[FeSCN²+ leq Because one mole of SCN is used up for each mole of FeSCN2+ ions produced, [SCN-Jeq can be determined by: [SCN-leq - [SCN-li-[FeSCN²+ leq = Knowing the values of [Feleqs [SCN-leqs and [FeSCN2+ legs you can now calculate the value of Ke the equilibrium constant. OBJECTIVE In this experiment, you will determine the equilibrium constant, Ke, for the following chemical reaction: MATERIALS Fe³+ (aq) + SCN (aq) →→→ FeSCN²+ (aq) iron(III) thiocyanate thiocyanoiron (III) Vernier LabQuest LabQuest App Vernier Colorimeter 6 plastic cuvettes five 20 x 150 mm test tubes three 100 mL beakers thermometer PROCEDURE 1. Obtain and wear goggles. 2. Label four 20 x 150 mm test tubes 1-4. Pour about 30 mL of 0.0020 M Fe(NO3)3 into a clean, dry 100 ml beaker. Pipet 5.0 mL of this solution into each of the four labeled test tubes. Use a pipet pump or bulb to pipet all solutions. CAUTION: Fe(NO)s solutions in this experiment are prepared in 1.0 M HNO, and should be handled with care. Pour about 25 mL of the 0.0020 M KSCN into another clean, dry 100 mL beaker. Pipet 2, 3, 4 and 5 mL of this solution into Test Tubes 1-4, respectively. Obtain about 25 mL of distilled water in a 100 ml beaker. Then pipet 3, 2, 1 and 0 mL of distilled water into Test Tubes 1-4, respectively, to bring the total volume of each test tube to 10 mL. Mix each solution thoroughly with a stirring rod. Be sure to clean and dry the stirring rod after each mixing. Measure and record the temperature of one of the above solutions to use as the temperature for the equilibrium constant, Ke. Volumes added to each test tube are summarized below: Test Tube Number 1 2 3 4 Fe(NO3) (mL) 20-2 5 0.0020 M KSCN 0.0020 M Fe(NO3)3 (in 1.0 M HNO3) 0.200 M Fe(NO3)3 (in 1.0 M HNO3) four pipets pipet bulb or pipet pump tissues (preferably lint-free). 5 5 5 KSCN (mL) 2 3 4 5 H₂O (mL) 3. Prepare a standard solution of FeSCN2+ by pipetting 18 mL of 0.200 M Fe(NO3)3 into a 20x150 mm test tube labeled "5". Pipet 2 mL of 0.0020 M KSCN into the same test tube. Stir thoroughly. • Wipe the outside of each cuvette with a lint-free tissue. • Handle cuvettes only by the top edge of the ribbed sides. 3 2 1 0 4. Prepare a blank by filling a cuvette 3/4 full with distilled water. To correctly use cuvettes, remember: • Dislodge any bubbles by gently tapping the cuvette on a hard surface. Always position the cuvette so the light passes through the clear sides. Chemistry with Vernier Chemical Equilibrium: Finding a Constant, Ke 5. Connect the Colorimeter to LabQuest and choose New from the File menu. 6. Calibrate the Colorimeter. a. Place the blank in the cuvette slot of the Colorimeter and close the lid. b. Press the button on the Colorimeter to select a wavelength of 470 nm. Press the CAL button on the Colorimeter. When the LED stops flashing, the calibration is complete. 7. Set up the data collection mode. a. On the Meter screen, tap Mode. Change the mode to Events with Entry. b. Enter the Name (Concentration) and Units (mol/L). Select OK. 8. You are now ready to collect absorbance data for the four equilibrium systems and the standard solution. a. With the Test Tube 1 solution, fill a new cuvette 3/4 full, and place it in the device. b. When the value displayed on the screen has stabilized, tap Keep and enter 1 (the trial number). Select OK. The absorbance and concentration values have now been saved for the first solution. Write the absorbance value on the report sheet for trial 1. c. With the Test Tube 1 solution, fill a new cuvette 3/4 full with Test Tube 2 solution, and place it in the device. Press run twice, and then "append". After the reading stabilizes, tap Keep and enter 2. Select OK to continue. Again, record the absorbance value for Trial 2, in your data table. d. Repeat the procedure to find the absorbance of the solutions in Test Tubes 3, 4, and 5 (the standard solution) with a new cuvette for each. Record the absorbance values, for each Trial, in your data table. e. Stop data collection. f. Examine the data points on the displayed graph. YOU SHOULD HAVE A STRAIGHT LINE INCREASING UP TO THE RIGHT. If you do not please contact your instructor!! g. If you want to check any data values, tap any data point. As you tap a data point, the absorbance and concentration values are displayed to the right of the graph. h. Dispose of all solutions in the waste container. Cuvettes and pipettes are also disposable. Lean any Sharpie off of the glassware.. PROCESSING THE DATA 1. Write the K expression for the reaction in the Data and Calculation table. 2. Calculate the initial concentration of Fe³+, based on the dilution that results from adding KSCN solution and water to the original 0.0020 M Fe(NO3)3 solution. See Step 2 of the procedure for the volume of each substance used in Trials 1-4. Calculate [Fe3+]; using the equation: Fe(NO3)3 mL [Feli= total mL This should be the same for all four test tubes. × (0.0020 M) LabQuest 20 3. Calculate the initial concentration of SCN", based on its dilution by Fe(NO3)3 and water: KSCN mL [SCN-li total ml × (0.0020 M) In Test Tube 1, [SCN-];-(2 mL/10 mL)(0.0020 M) = 0.00040 M. Calculate this for the other three test tubes. 4. [FeSCN2Jeq is calculated using the formula: Aeg X Astd x [FeSCN²+]std [FeSCN2 Jeq where Aeq and Asd are the absorbance values for the equilibrium and standard test tubes, respectively, and [FeSCN2 Isid (1/10)(0.0020) = 0.00020 M. Calculate [FeSCN²leq for each of the four trials. 5. [Feleq: Calculate the concentration of Fe³+ at equilibrium for Trials 1-4 using the equation: [Fe³ leq=[Fe³+]-[FeSCN²* Jeq 6. [SCN-Jeq: Calculate the concentration of SCN- at equilibrium for Trials 1-4 using the equation: [SCN-Jeq [SCN-]-[FeSCN2+ leq 7. Calculate K for Trials 1-4. Be sure to show the Ke expression and the values substituted in for each of these calculations. 8. Using your four calculated K, values, determine an average value for Ke. How constant were your Ke values? DATA AND CALCULATIONS (2 PTS. PER ENTRY UNLESS OTHERWISE MARKED) Trial 1 1.25 Absorbance of standard (Trial 5) Absorbance K. expression (3 pt) [Fe] [SCN-] [FeSCN²] [Fe] [SCN-Jea K value (3 pt ea.) Average of K, values (3 pt) Chemistry with Vernier Chemical Equilibrium: Finding a Constant, Ke Trial 2 1.66 0.58 Ke= Trial 3 2.07 Temperature Trial 4 2.35 32.5 c 76 °C 20-5 LabQuest 20 1. (5 pt.) Should the equilibrium constant be around the same for all of the test solution Explain. 2. (5 pt.)How closely did your equilibrium constants match each other, and what experimental factors might cause them to be different?
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate the equilibrium constant Kc for the following reaction from standard electrode potentials. Fe(s) + Sn**(ag) = Fe* (ag) + Sn*(aq)
-
The purpose of this assignment is to make sure that the students can work with the fork, pipe, and dup system calls. It should also grant some insight into how the shell makes input/output...
-
The purpose of this mini-project is to help you verify the Rule for Sample Means, using a physical simulation. Suppose you are interested in measuring the average amount of blood contained in the...
-
Consider again examples 9.2 and 9.3 , and once again assume that there are two states of the world for each random variable, denoted by V L j , V H j and n L , n H . Denote the probabilities for...
-
The EPA has increased its BACT standards to impose greenhouse gas emissions standards for passenger cars, light-duty trucks, and medium-duty passenger vehicles and all businesses that emit greenhouse...
-
Suppose the child runs with a different initial speed, but that everything else in the system remains the same. What initial speed does the child have if the angular speed of the system after the...
-
Almetals, Inc., a Michigan company, entered into a contract with the German firm Wickeder Westfalenstahl regarding the purchase of clad metal, a specialty metal used in a variety of industries but...
-
The following are five independent situations. 1. The audit firm, Weaver and Jones, LLP, received a subpoena for its documentation related to the audit of Westbrook Corporation's financial...
-
discuss the advantages and disadvantages of Database Management Systems (DBSMs). What do you personally think of Database Management Systems?
-
In cryptography, cipher text is encrypted or encoded text that is unreadable by a human or computer without the proper algorithm to decrypt it into plain text. The impact of erroneous cipher texts on...
-
A,B,C three points on a like L a = segment AC, b = segment BC with a>b M the midpoint of AB and n the line perpendicular to L passing through B the circle (e) with center M and radius MC. D and E...
-
What connecting the topic directly to the listeners is known?
-
A parallel plate that has an area of 1 3 . 5 cm 2 with the 2 plates being 8 . 9 mm apart, is hooked up to a 9 . 0 V battery. a . What is the capacitance of the capacitor? b . What is the charge on...
-
Determine the most valuable aspect of this course to you and explain how you plan to apply it to your personal financial practices.Share two ways that you feel you will be able to apply the concepts...
-
2. Compare a Magnetic Drive Sealless Pump with a Canned Pump. a. Discuss one common feature of both pumps [1m] b. Discuss three differences between these 2 pump types [3m]
-
that the company should report in balance sheet. 8 . Pluto Corporation purchased inventory for$ 1 0 1 , 1 0 0 . The company plans to sell it at $ 1 0 3 , 3 0 0 , but estimates that it will incur...
-
2) Show only one iteration for solving the following problem by using dual simplex method? p.) (There should be two tables in your paper) Min.z 3x1+ 4x2 + 5x3 x1 + 2x2+3x3 >= 5 2x1 + 2x2 + x3 >=6 x1,...
-
Banner Company acquires an 80% interest in Roller Company for $640,000 cash on January 1, 2013. The NCI has a fair value of $160,000. Any excess of cost over book value is attributed to goodwill. To...
-
To test the effects of drugs and alcohol use on driving performance, 20 volunteers were each asked to take a driving test under three conditions: sober, after two drinks, and after smoking marijuana....
-
Here is a potential survey question: Studies have shown that consuming one alcoholic drink daily helps reduce heart disease. How many alcoholic drinks do you consume daily? a. Explain which two of...
-
Remember that the probability that a birth results in a boy is about .51. You offer a bet to an unsuspecting friend. Each day you will call the local hospital and find out how many boys and how many...
-
Consider a Lagrangian \(L^{\prime}=L+d f / d t\), where the Lagrangian is \(L=\) \(L\left(q_{k}, \dot{q}_{k}, tight)\), and the function \(f=f\left(q_{k}, tight)\). (a) Show that...
-
Show that the function \(L^{\prime}\) given in the preceding problem must obey Lagrange's equations if \(L\) does, directly from the principle of stationary action. Lagrange's equations do not have...
-
In Example 4.8 we analyzed the case of a bead on a rotating parabolic wire. The energy of the bead was not conserved, but the Hamiltonian was: There is an equilibrium point at \(r=0\) which is...

Study smarter with the SolutionInn App


