The specific heat of ethyl alcohol (C2H5OH) is 2.427J/g*c, it's boiling point is 78.3*c at 1.00atm...
Fantastic news! We've Found the answer you've been seeking!
Question:
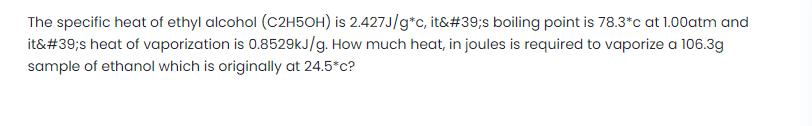
Transcribed Image Text:
The specific heat of ethyl alcohol (C2H5OH) is 2.427J/g*c, it's boiling point is 78.3*c at 1.00atm and it's heat of vaporization is 0.8529kJ/g. How much heat, in joules is required to vaporize a 106.3g sample of ethanol which is originally at 24.5*c? The specific heat of ethyl alcohol (C2H5OH) is 2.427J/g*c, it's boiling point is 78.3*c at 1.00atm and it's heat of vaporization is 0.8529kJ/g. How much heat, in joules is required to vaporize a 106.3g sample of ethanol which is originally at 24.5*c?
Expert Answer:
Answer rating: 100% (QA)
1 Calculate the change in temperature of the sample T 783C 245C 538C 2 Calcul... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
BOB'S BACK HOES STUDENT EXERCISE Overview In this case-study, you, the student, will set up a new service company file using the Easy Step Interview found by clicking on Detailed Start after clicking...
-
Q.1. Let's practice the concept of polyphase decomposition of a sequence. Consider the following sequence: [10] CLO 3 h[n] = 2n + 1 0ns7 a) Consider M = 4 and pass h[n] through the block diagram...
-
Ethyl alcohol has about half the specific heat of water. If equal-mass samples of alcohol and water in separate beakers are supplied with the same amount of energy, compare the temperature increases...
-
Write the formulas of these compounds: sulfur trioxide; phosphorus pentachloride; dinitrogen tetroxide.
-
Silver Aviation assembles small aircraft for commercial use. The majority of its business is with small freight airlines serving areas whose airports do not accommodate larger planes. The remainder...
-
Use Laplace transforms to solve the initial value problems in Problems 27 through 38. x (3) + x" - 6x' = 0; x (0) = 0, x'(0) = x" (0) = 1
-
For each of the situations listed, identify the primary standard from the IMA Statement of Ethical Professional Practice that is violated (competence, confidentiality, integrity, or credibility)....
-
Fred and Frieda have always wanted to enter the blueberry business. They locate a 50- acre piece of hillside in Maine that is covered with blueberry bushes. They figure that the annual yield from the...
-
1. In connection with the circuit drawn below, the value of current flowing through 2 kQ resistor is 10-4A. 2. 5V 1 www 2. 3. 4. 5. 6. 7. An audio signal Vm = 20 sin 2(1500t) amplitude modulates a...
-
On January 1, Year 1, the Vine Company purchased 60,000 of the 80,000 ordinary shares of the Devine Company for $80 per share. On that date, Devine had ordinary shares of $3,500,000, and retained...
-
Henry is a carrier of Huntington's Disease. His partner Sophie has a sister with Huntington's Disease, which means the probability Sophie is a carrier is 2/3. If Sophie is a carrier, each child she...
-
According to economists Henry Saffer of Kean University, Frank J. Chaloupka of the University of Illinois at Chicago, and Dhaval Dave of CUNY Graduate Center, using the criminal justice system to...
-
Locate at least one article that comments on the tax provisions for capital gains that were included as part of the 2012 Taxpayer Relief Act. Summarize the comments and provide a citation for your...
-
Soft-drink companies pay universities for the exclusive pouring rights to sell their products on campus. In a recent deal, UCLA signed a contract with Pepsi for $1.5 million per year limiting...
-
Over what ranges of taxable income in 2017 will the total income tax liability for two persons with equal incomes who file as single individuals equal their income tax liability if they file jointly...
-
The title of an article in The Wall Street Journal was Pricing of Products Is Still an Art, Often Having Little Link to Costs. In the article, the following cases were cited: Vodka pricing: All...
-
The following is the trend of PSEI monthly values: Axis Title 1. 1,600.00 1,400.00 1,200.00 1,000.00 800.00 600.00 400.00 200.00 0.00 Required: PSEI y= 5.0896x+614.29 1 6...
-
The MIT Sloan School of Management is one of the leading business schools in the U.S. The following table contains the tuition data for the masters program in the Sloan School of Management. a. Use...
-
Create graphs similar to Fig. 14-5, but for lead and ethyl alcohol. Compare and contrast them with each other and with the graph for water. Are there any temperature ranges for which all three...
-
What is the pressure inside a 38.0-L container holding 105.0 kg of argon gas at 21.6C?
-
Can the temperature of a system remain constant even though heat flows into or out of it? If so, give examples.
-
The file ApplianceShipments.csv contains the series of quarterly shipments (in million dollars) of US household appliances between 1985 and 1989 (data courtesy of Ken Black). a. Create a...
-
Canadian Manufacturing Workers Workhours. The time plot in Figure 17.9 describes the average annual number of weekly hours spent by Canadian manufacturing workers (data are available in...
-
Shampoo Sales. The file ShampooSales.csv contains data on the monthly sales of a certain shampoo over a three-year period. [Source: Hyndman and Yang (2018). a. Create a well-formatted time plot of...

Study smarter with the SolutionInn App


