Question: The student took the same 25 mL flask and filled the entire volume of the flask with an unknown liquid. The mass of the
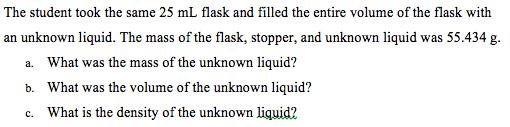
The student took the same 25 mL flask and filled the entire volume of the flask with an unknown liquid. The mass of the flask, stopper, and unknown liquid was 55.434 g. a. What was the mass of the unknown liquid? b. What was the volume of the unknown liquid? c. What is the ensity of the unknown liquid?
Step by Step Solution
3.34 Rating (148 Votes )
There are 3 Steps involved in it
To solve these questions we need some additional data from the experiment specifically the mass of t... View full answer

Get step-by-step solutions from verified subject matter experts


