The synthesis of the benzyl proceeds as follows. it is purified by means of crystallization from ethanol.
Fantastic news! We've Found the answer you've been seeking!
Question:
The synthesis of the benzyl proceeds as follows.
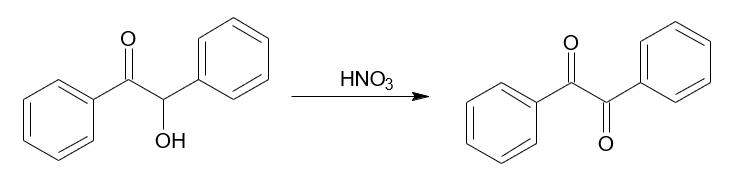 it is purified by means of crystallization from ethanol.
it is purified by means of crystallization from ethanol.
1. Why is ethanol used to purify benzyl?
2. What impurities are eliminated in the crystallization process? (reagents and by-products)
3. Explain the solubility of the product (benzyl) and the impurities in hot and cold ethanol
4. Is it correct to use ethanol for crystallization? Can you use another solvent, or a mixture of solvents?
Related Book For 

Probability and Statistics for Engineering and the Sciences
ISBN: 978-1305251809
9th edition
Authors: Jay L. Devore
Posted Date:




