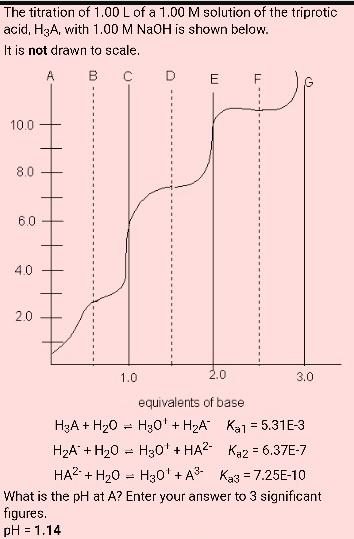
The titration of 1.00 L of a 1.00 M solution of the triprotic acid, H3A, with...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The titration of 1.00 L of a 1.00 M solution of the triprotic acid, H3A, with 1.00 M NaOH is shown below. It is not drawn to scale. A B 10.0 8.0 6.0 4.0 2.0 H3A + H₂0 H₂A + H₂0 1.0 = = D = E 2.0 equivalents of base H30' + H₂A" H30' + HA2 F 3.0 Ka1 = 5.31E-3 Kaz = 6.37E-7 HA² + H₂O H₂0¹ + A³ Ka3 = 7.25E-10 What is the pH at A? Enter your answer to 3 significant figures. pH = 1.14 What is the pH at G? Enter your answer to 3 significant figures. pH = The titration of 1.00 L of a 1.00 M solution of the triprotic acid, H3A, with 1.00 M NaOH is shown below. It is not drawn to scale. A B 10.0 8.0 6.0 4.0 2.0 H3A + H₂0 H₂A + H₂0 1.0 = = D = E 2.0 equivalents of base H30' + H₂A" H30' + HA2 F 3.0 Ka1 = 5.31E-3 Kaz = 6.37E-7 HA² + H₂O H₂0¹ + A³ Ka3 = 7.25E-10 What is the pH at A? Enter your answer to 3 significant figures. pH = 1.14 What is the pH at G? Enter your answer to 3 significant figures. pH =
Expert Answer:
Answer rating: 100% (QA)
The no of moles of acid 1 L 1 molL 1 mol and the no of mol... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Let A, B be sets. Define: (a) the Cartesian product (A B) (b) the set of relations R between A and B (c) the identity relation A on the set A [3 marks] Suppose S, T are relations between A and B, and...
-
Shown here are condensed income statements for two different companies (both are organized as LLCs and pay no income taxes). Required 1. Compute times interest earned for Ace Company. 2. Compute...
-
On December 31, 2014, Firth Company borrowed $62,092 from Paris Bank, signing a 5-year, $100,000 zero-interest-rate note. The note was issued to yield 10% interest. Unfortunately, during 2016, Firth...
-
A stirred-tank blending system with a bypass stream is shown in Figure. The control objective is o control the composition of a key component in the exit stream, x4. The chief disturbance variables...
-
At what radial distance \(r\) from the center of a toroid of 200 windings does the magnitude of the magnetic field equal that found inside a solenoid that has 500 turns per meter of length? Assume...
-
The following information pertains to Boone Company for 2009. Beginning inventory 70 units @ $26 Units purchased 280 units @ $30 Ending inventory consisted of 30 units. Boone sold 320 units at $40...
-
Trying to break an encryption key by trying every possible combination of characters is called what? 1 point A social engineering attack A brute force attack A rainbow table attack A known cyphertext...
-
The following selected accounts appear on the Balance Sheet of Examination Corporation as of December 31, 2021. Stockholders' Equity Preferred 5% Stock, $100 par (100,000 shares authorized, 25,000...
-
Sangria is a US-based company whose products aim to promote happy, low-stress lifestyles. Let's calculate the WACC of Sangria. Its accounting and market value balances are: Value in books Asset value...
-
A gas engine working on 4-stroke cycle has a cylinder diameter \(300 \mathrm{~mm}\) and stroke length of 500 \(\mathrm{mm}\) is running at \(220 \mathrm{rpm}\). Its mechanical efficiency is \(80 \%\)...
-
A furnace wall consists of \(100 \mathrm{~mm}\) thick refractory brick and \(800 \mathrm{~mm}\) thick insulating firebrick separated by an air gap. The outside wall is covered with a \(20...
-
Visit the social bookmarking site you joined and look up a topic of interest for you. Select ten headlines that have been saved by other users. Classify the headlines according to the type of...
-
A steel pipe of \(100 \mathrm{~mm}\) bore and \(10 \mathrm{~mm}\) wall thickness, carrying steam at \(250^{\circ} \mathrm{C}\), is insulated with \(30 \mathrm{~mm}\) of a molded high-temperature...
-
A small hemispherical oven is built of an inner layer of insulating firebrick \(110 \mathrm{~mm}\) thick, and an outer covering \(80 \%\) magnesia \(45 \mathrm{~mm}\) thick. The inner surface of the...
-
Amazon.com, Inc.'s financial statements are presented in Appendix D. Financial statements of Wal-Mart Stores, Inc. are presented in Appendix E. Instructions for accessing and using the complete...
-
Solve each problem. Find the coordinates of the points of intersection of the line y = 2 and the circle with center at (4, 5) and radius 4.
-
Solving the following road-map problem depends on determining the structure of A, the key intermediate. Give structures for compounds A through K. hept-1-yne SOCI CH3CuLi (2) (CHj,S (2) Ho HCN (2)...
-
Predict the products formed when m-cresol (m-methylphenol) reacts with (a) NaOH and then ethyl bromide (b) Acetyl chloride, (c) Bromine in CCl4 the dark (d) Excess bromine in CCl4 the light (e)...
-
(a) Draw the structure of the transition state for the second propagation step in the chlorination of methane. CH3 + Cl2 CH3Cl + Cl Show whether the transition state is product-like or...
-
In one cell, gene C is expressed, whereas in another cell, gene C is inactive. After the cells are fused experimentally, both copies of gene C are expressed. This observation could be explained by a....
-
During the process of XCI, which chromosome expresses the Xist gene and which one expresses the Tsix gene? a. Xist is expressed only by Xa, and Tsix is expressed only by Xi. b. Xist is expressed only...
-
A form of chromatin that silences gene expression and varies in location among different types of cells is a. euchromatin. b. constitutive heterochromatin. c. facultative heterochromain. d. Both b...

Study smarter with the SolutionInn App


