The titration technique is used to do the reaction because the 0.1 M NaOH can be added
Fantastic news! We've Found the answer you've been seeking!
Question:
The titration technique is used to do the reaction because the 0.1 M NaOH can be added sequentially and the pH is monitored as the moles of HA react to form NaA. The Ka is calculated at the three lettered points. Use the information is question 1 to find:
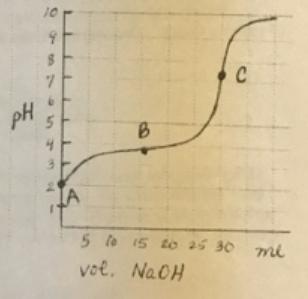
(a) How many moles of HA are present at point A (initial)?
(b) How many moles of HA are present at point B (the half-equivalence point)?
(c) How many moles of HA are present at point C (the equivalence point)?
Related Book For 

Probability & Statistics for Engineers & Scientists
ISBN: 978-0130415295
7th Edition
Authors: Ronald E. Walpole, Raymond H. Myers, Sharon L. Myers, Keying
Posted Date:




