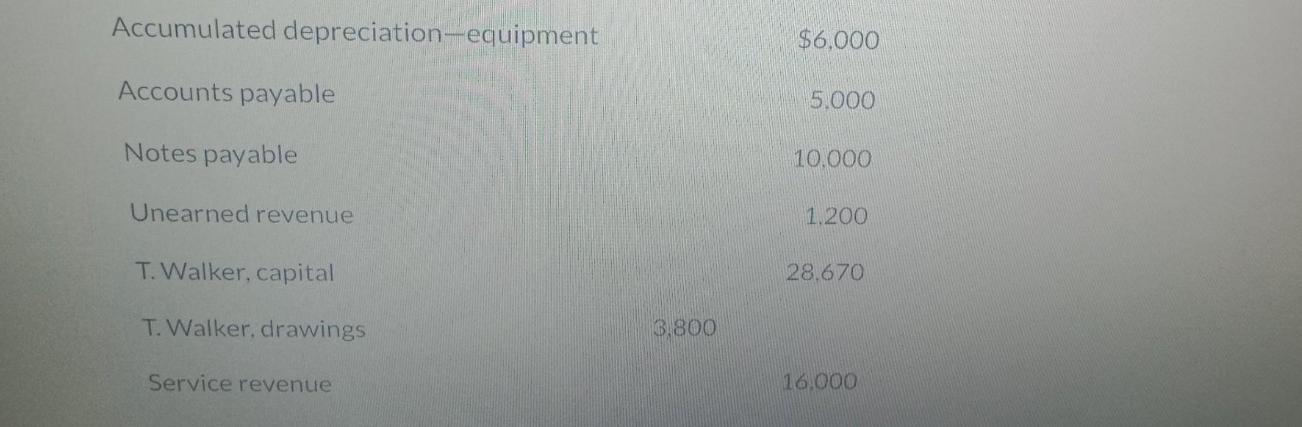
The unadjusted trial balance for Sandhill Designs at its year end, April 30, 2024, is as...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The unadjusted trial balance for Sandhill Designs at its year end, April 30, 2024, is as follows: Cash Accounts receivable Prepaid rent SANDHILL DESIGNS Trial Balance April 30, 2024 Equipment Debit $12,000 8.200 5.500 24.000 Credit Accumulated depreciation-equipment Accounts payable Notes payable Unearned revenue T. Walker, capital T. Walker, drawings Service revenue 3.800 $6.000 5.000 10.000 1.200 28.670 16.000 Service revenue Salaries expense Interest expense Depreciation expense Additional information: 9,700 920 2,750 $66,870 16,000 $66,870 1. 2. 3. $560 of the unearned revenue is to be recognized because the related gardening services have been provided by April 30, 2024. The equipment has an estimated useful life of eight years. Interest on the note payable is due on the first day of each month for the previous month's interest. The note payable has a 6% annual interest rate. Required Prepare adjusting entries for the month ended April 30,2024 Post the adjusting entries in T-Accounts The unadjusted trial balance for Sandhill Designs at its year end, April 30, 2024, is as follows: Cash Accounts receivable Prepaid rent SANDHILL DESIGNS Trial Balance April 30, 2024 Equipment Debit $12,000 8.200 5.500 24.000 Credit Accumulated depreciation-equipment Accounts payable Notes payable Unearned revenue T. Walker, capital T. Walker, drawings Service revenue 3.800 $6.000 5.000 10.000 1.200 28.670 16.000 Service revenue Salaries expense Interest expense Depreciation expense Additional information: 9,700 920 2,750 $66,870 16,000 $66,870 1. 2. 3. $560 of the unearned revenue is to be recognized because the related gardening services have been provided by April 30, 2024. The equipment has an estimated useful life of eight years. Interest on the note payable is due on the first day of each month for the previous month's interest. The note payable has a 6% annual interest rate. Required Prepare adjusting entries for the month ended April 30,2024 Post the adjusting entries in T-Accounts
Expert Answer:
Answer rating: 100% (QA)
Prepare the adjusting entries for the month ended April 30 2024 Adjusting entry to recognize the ear... View the full answer

Related Book For 

Accounting Principles Volume 1
ISBN: 9781119786818
9th Canadian Edition
Authors: Jerry J. Weygandt, Donald E. Kieso, Paul D. Kimmel, Barbara Trenholm, Valerie Warren, Lori Novak
Posted Date:
Students also viewed these accounting questions
-
This is a famous problem from probability, based on the old game show Let's Make a Deal and its host, Monty Hall. There are many, many explanations of this problem on the internet. Feel free to read...
-
The unadjusted trial balance for Garden Designs at its year end, April 30, 2017, is as follows: Additional information: 1. $500 of the unearned revenue has been earned by April 30, 2017. 2. The...
-
What key components of a business model are very important in an enterprise?
-
With respect to strategies used by land conservation groups to preserve land, conservation easements seem to be expanding more rapidly than buying land for preservation. In what respect might...
-
A .22 rifle bullet, traveling at 350 m/s, strikes a large tree, which it penetrates to a depth of 0.130 m. The mass of the bullet is 1.80 g. Assume a constant retarding force. (a) How much time is...
-
What environmental factors must be considered in analyzing companies?
-
Are conventional financial accounting and external ecological accounting complements to each other or substitutes for each other? Discuss.
-
Below are transactions for Hurricane Company during 2015. a. On October 1, 2015, Hurricane lends $10,000 to another company. The other company signs a note indicating principal and 8% interest will...
-
Valley Designs issued a 120-day, 4% note for $76,800, dated April 10 to Bork Furniture Company on account. Required: a. Determine the due date of the note. b. Determine the maturity value of the...
-
When you think of political persuasion, you may think of the effortsthat political campaigns undertake to persuade you that their candidate is betterthan the other candidate. In truth, campaigns are...
-
A company's actual assessable payroll is $735,250.00. The premium rate is $1.35 per $100 of assessable payroll. Calculate the company's annual worker's compensation assessment.
-
List some design and implementation issues faced, when implementing the corruption pattern. Explain each issue.
-
How are the above terms associated with our EBT of competition? List possible application domains for the SAP competition.
-
Are there any constraints that need to be handled by the competition pattern?
-
List three challenges in formulating the trust analysis pattern.
-
T/F: Traditional analysis patterns have limited reuse. Why?
-
Suppose that in Country A only two goods are produced, which are books and textiles. The CPI basket includes 30 units of books and 60 units of textiles. The prices of these goods in 2019 and 2020 are...
-
Write the binomial probability in words. Then, use a continuity correction to convert the binomial probability to a normal distribution probability. P(x 110)
-
Kari Downs, an auditor with Wheeler & Co., CPAs, is performing a review of Depue Companys inventory account. Depue did not have a good year, and top management is under pressure to boost reported...
-
A list of accounts and their balances of ONeills Psychological Services, at its year end July 31, 2024, is presented below. Instructions a. Prepare a trial balance in financial statement order. b....
-
A new accountant at Magenta Company is trying to identify which of the following amounts should be reported as the current asset Cash and Cash Equivalents in the year-end balance sheet, as at June...
-
Clausius-Clayperon equation is applicable to (a) Solid-vapour (b) Solid-liquid (c) Liquid-vapour (d) All of these.
-
The reaction \(A(\mathrm{l}) ightarrow R(\mathrm{~g})\) is allowed to reach equilibrium condition in an autoclave. At equilibrium there are two phases - one a pure liquid phase of \(A\) and the other...
-
The equilibrium constant for the reaction \(\mathrm{N}_{2}(\mathrm{~g})+3 \mathrm{H}_{2}(\mathrm{~g}) ightarrow 2 \mathrm{NH}_{3}\) is 0.1084 . Under the same conditions, the equilibrium constant for...

Study smarter with the SolutionInn App