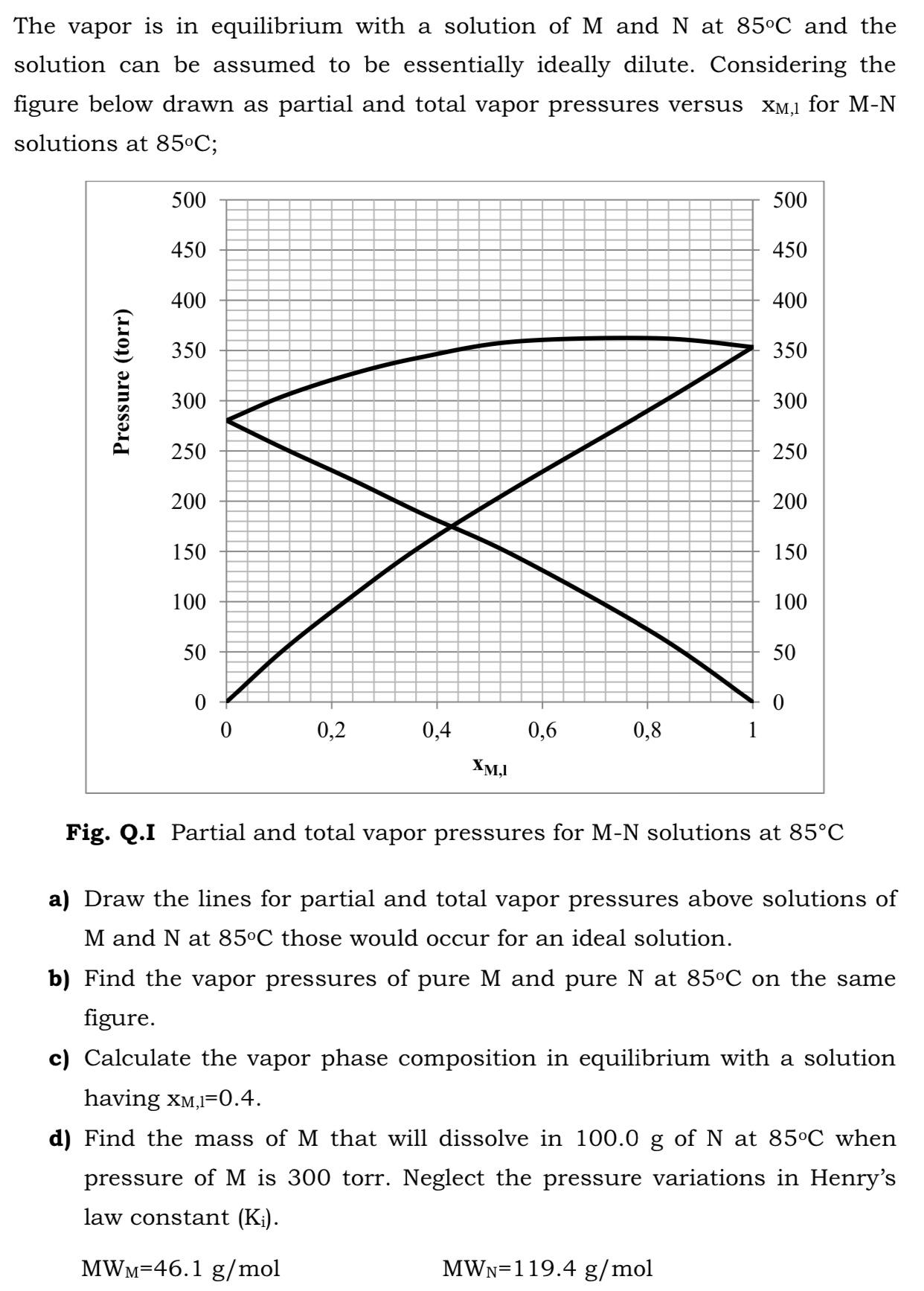
The vapor is in equilibrium with a solution ofM and N at 85C and the solution...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The vapor is in equilibrium with a solution ofM and N at 85°C and the solution can be assumed to be essentially ideally dilute. Considering the figure below drawn as partial and total vapor pressures versus XM,1 for M-N solutions at 85°C; 500 500 450 450 400 400 350 350 300 300 250 250 200 200 150 150 100 100 50 50 0,2 0,4 0,6 0,8 XM,I Fig. Q.I Partial and total vapor pressures for M-N solutions at 85°C a) Draw the lines for partial and total vapor pressures above solutions of M and N at 85°C those would occur for an ideal solution. b) Find the vapor pressures of pure M and pure N at 85°C on the same figure. c) Calculate the vapor phase composition in equilibrium with a solution having xM,I=0.4. d) Find the mass of M that will dissolve in 100.0 g of N at 85°C when pressure of M is 300 torr. Neglect the pressure variations in Henry's law constant (K¡). MWM=46.1 g/mol MWN=119.4 g/mol Pressure (torr) The vapor is in equilibrium with a solution ofM and N at 85°C and the solution can be assumed to be essentially ideally dilute. Considering the figure below drawn as partial and total vapor pressures versus XM,1 for M-N solutions at 85°C; 500 500 450 450 400 400 350 350 300 300 250 250 200 200 150 150 100 100 50 50 0,2 0,4 0,6 0,8 XM,I Fig. Q.I Partial and total vapor pressures for M-N solutions at 85°C a) Draw the lines for partial and total vapor pressures above solutions of M and N at 85°C those would occur for an ideal solution. b) Find the vapor pressures of pure M and pure N at 85°C on the same figure. c) Calculate the vapor phase composition in equilibrium with a solution having xM,I=0.4. d) Find the mass of M that will dissolve in 100.0 g of N at 85°C when pressure of M is 300 torr. Neglect the pressure variations in Henry's law constant (K¡). MWM=46.1 g/mol MWN=119.4 g/mol Pressure (torr)
Expert Answer:
Answer rating: 100% (QA)
a The experimental and Idea l partial vapour pressures of Solu... View the full answer

Related Book For 

Foundations of Financial Management
ISBN: 978-1259194078
15th edition
Authors: Stanley Block, Geoffrey Hirt, Bartley Danielsen
Posted Date:
Students also viewed these chemical engineering questions
-
When returns from a project can be assumed to be normally distributed, such as those shown in Figure (represented by a symmetrical, bell-shaped curve), the areas under the curve can be determined...
-
Soil water with 10^-3 N alkalinity is in equilibrium with a soil gas in which PCO2 = 10^-1 atm. Calculate the pH and total dissolved inorganic carbon concentration (M).
-
A water is in equilibrium with Ca3(PO4)2(s). The solution contains other cations and anions, but no other weak acids or bases or sources of Ca and PO4, and has a pH of 8.6. Ignore complexes. (a) What...
-
Explain these points in detail w.r.t application of control in auditing with examples. 1: Relate data capture control with internal control in auditing with example 2: Relate validation control with...
-
Master Mind Games, Inc. is a new corporation started on January 1, 2015. The following transactions occurred during the first year of operations. a. On January 1, 2015, the owners invested a total of...
-
The average ticket price P at movie theaters from 2001 through 2014 can be approximated by the model P = 0.0038t2 + 0.272t + 5.25, 1 t 14 where t represents the year, with t = 1 corresponding to...
-
Draw a diagram showing the developmental steps of one of the projects you recently designed. Which development model did you follow (waterfall, spiral, etc.)?
-
In 2014, the Northwest Ballet Association (NBA), a not-for-prot performing arts organization, undertook a major capital campaign to fund a new theater, expected to cost $10 million. It was quickly...
-
Please summarize the analysis For the year ending December 31, 2018, the Gross Revenue Index is 0, which means there was no gross revenue in 2018 or it remained the same as the base year (2017). The...
-
What challenges do health care facilities face in addressing ergonomic job design for nurses and nursing assistants? How can meeting these challenges benefit these facilities?
-
"The Bank of Canada can perfectly control the amount of the monetary base, but has less control over the composition of the monetary base." Is this statement true, false, or uncertain? Explain your...
-
What limits are placed on employment related expenses for the household and dependent care credit?
-
Explain the circumstances that permit a shareholder, who receives an installment obligation from a liquidating corporation, to defer recognition of gain until the shareholder receives the payments on...
-
What are the seven tests that help to establish material participation?
-
How many years of post-secondary education expenses are eligible for the Hope tuition credit?
-
What are the most common types of qualified plans for small businesses?
-
1. What class of airport must large air carriers operate in the United States? 2. Name five categories of information the Airport Certification Manual must contain. 3. List three considerations when...
-
Write a declaration for each of the following: a. A line that extends from point (60, 100) to point (30, 90) b. A rectangle that is 20 pixels wide, 100 pixels high, and has its upper-left corner at...
-
How did the SarbanesOxley Act impact corporations financial reports?
-
Betty Bronson has just retired after 25 years with the electric company. Her total pension funds have an accumulated value of $180,000, and her life expectancy is 15 more years. Her pension fund...
-
Assume a firm has earnings before depreciation and taxes of $440,000 and depreciation of $140,000. a. If it is in a 35 percent tax bracket, compute its cash flow. b. If it is in a 20 percent tax...
-
On a rifle that has a telescopic sight, the telescope is usually not parallel to the barrel of the rifle. The angle the telescope makes with the barrel has to be adjusted for the distance to the...
-
A package is dropped from a helicopter traveling at \(15 \mathrm{~m} / \mathrm{s}\) (horizontally) at an altitude of \(200 \mathrm{~m}\), but the parachute attached to the package fails to open. (a)...
-
The velocity of an object is given in SI units by \(\vec{v}=\left(a t-b t^{2} ight) \hat{\imath}+c \hat{\jmath}\), with \(a=14 \mathrm{~m} / \mathrm{s}^{2}, b=10 \mathrm{~m} / \mathrm{s}^{3}\), and...

Study smarter with the SolutionInn App


