The wavefunction of an electron in an eigenstate of a Hydrogen atom is given by 3/2...
Fantastic news! We've Found the answer you've been seeking!
Question:

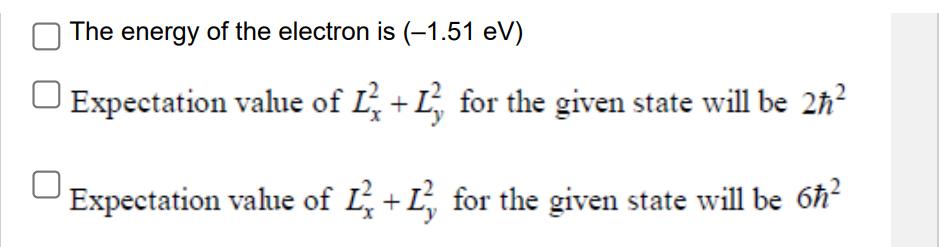
Transcribed Image Text:
The wavefunction of an electron in an eigenstate of a Hydrogen atom is given by 3/2 1 1 r LAD (2) exp cos ao ao 240 y (r,0,0) = 4√ r where ao is the first Bohr radius. Which of the following statement(s) is/are CORRECT? The energy of the electron is (-3.4 eV) The energy of the electron is (-1.51 eV) | Expectation value of Ľ + L² for the given state will be 2ħ² Expectation value of L+L for the given state will be 6ħ² The wavefunction of an electron in an eigenstate of a Hydrogen atom is given by 3/2 1 1 r LAD (2) exp cos ao ao 240 y (r,0,0) = 4√ r where ao is the first Bohr radius. Which of the following statement(s) is/are CORRECT? The energy of the electron is (-3.4 eV) The energy of the electron is (-1.51 eV) | Expectation value of Ľ + L² for the given state will be 2ħ² Expectation value of L+L for the given state will be 6ħ²
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
A hydrogen atom is in its second excited state. Determine, according to quantum mechanics, (a) The total energy (in eV) of the atom, (b) The magnitude of the maximum angular momentum the electron can...
-
A hydrogen atom is in its second excited state, n = 3. Using the Bohr model of hydrogen, find (a) the linear momentum and (b) the angular momentum of the electron in this atom.
-
The de Broglie wavelength of an electron in a hydrogen atom is 1.66 nm. Identify the integer n that corresponds to its orbit.
-
Carey Company is borrowing $200,000 for one year at 12 percent from Second Intrastate Bank. The bank requires a 20 percent compensating balance. What is the effective rate of interest? What would the...
-
In the chapter, you learned that one way the government enforces agricultural price floors is to buy up the excess supply itself. If the government wanted to follow a similar kind of policy to...
-
Which of the four components of market assessment do you think are often most difficult to assess? Why?
-
Correlation between Residuals and Explanatory Variables. Consider a generic sequence of pairs of numbers \(\left(x_{1}, y_{1} ight), \ldots,\left(x_{n}, y_{n} ight)\) with the correlation coefficient...
-
On November 1, 2017, Nordin Inc. (Nordin) issued a $5,000,000 bond with a 6 percent coupon rate and a maturity date of October 31, 2022. Interest is paid semi-annually on April 30 and October 31. The...
-
1. Explain the transfer of energy from the water through the dam to become electricity that can be used in your home. ( In your response, be sure to identify sources of both potential and kinetic...
-
Does it surprise you that the CPA had to resort to reviewing court cases to find out if points are deductible? Does it surprise you that the correct answer differs depending what state you live in?...
-
1. what are the impacts of covid-19 on the foreign exchange market? 2. what are the effects of covid-19 on Balance of payment and how can it be mitigated?
-
Explain the relationship between the bankruptcy proceedings that many legacy airlines have entered into with respect to competition from the low-cost carriers.
-
Calculate the optimal markup on cost for each service, based on the following estimates of point price elasticity of demand: Passenger Type First Class Cost Elasticity $400 -1.25 -1.1 -3.5 -2.5...
-
What is the difference between the T-statistic and the F-statistic?
-
Focus on the airline industry: why is the upper portion of the kinked demand curve elastic and the lower portion inelastic?
-
What are the merits of using peak-load pricing at commercial airports?
-
In its fiscal 2022 annual report, Lowell Inc. reported cash of $732 million at year end. The statement of cash flows reports the following (in millions): Net cash flow from operating activities...
-
Graph one period of each function. y = 4 cos x
-
What proportions of ethanol and water should be mixed in order to produce 100 cm:' of a mixture containing 50 per cent by mass of ethanol? What change in volume is brought about by adding 1.00 cm3 of...
-
List the symmetry elements of the following molecules and name the point groups to which they belong: (a) Naphthalene, (b) Anthracene, (c) The three dichlorobenzenes.
-
Set up and solve the Huckel secular equations for the n electrons of NO-3. Express the energies in terms of the Coulomb integrals ao and aN and the resonance integral 13. Determine the delocalization...
-
A diploid organism has a total of 14 chromosomes and about 20,000 genes per haploid genome. Approximately how many genes are in each linkage group?
-
By conducting testcrosses, researchers have found that the sweet pea plant has seven linkage groups. How many chromosomes would you expect to find in leaf cells of sweet pea plants?
-
Describe the unique features of ascomycetes that facilitate genetic analysis of these fungi.

Study smarter with the SolutionInn App



