The % yield of ammonia as a function of time in the reaction: N 2 (g) +
Fantastic news! We've Found the answer you've been seeking!
Question:
The % yield of ammonia as a function of time in the reaction:
N2(g) + 3H2(g) ? 2NH3(g), ?H 1) is given below.
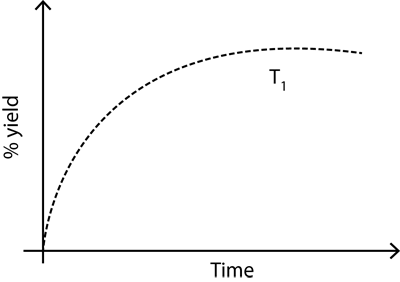
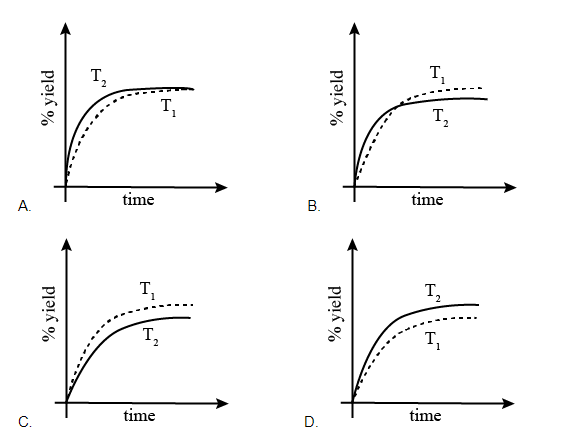
If this reaction is conducted at (P, T2), with T2 > T1, the %yield of ammonia as a function of time is represented by:
Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:




