Tila) transform to Ti() at 882 C, with a heat of transformation equal to 830 cal....
Fantastic news! We've Found the answer you've been seeking!
Question:
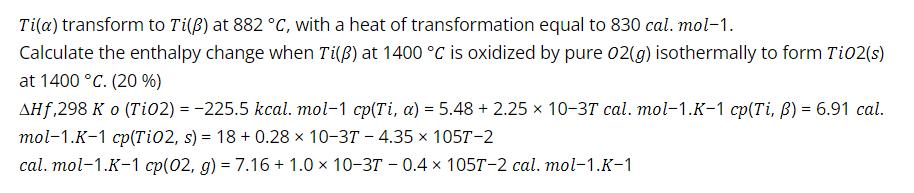
Transcribed Image Text:
Tila) transform to Ti(ß) at 882 °C, with a heat of transformation equal to 830 cal. mol-1. Calculate the enthalpy change when Ti(B) at 1400 °C is oxidized by pure 02(g) isothermally to form Ti02(s) at 1400 °C. (20 %) AHf,298 K o (Ti02) = -225.5 kcal. mol-1 cp(Ti, a) = 5.48 + 2.25 x 10-3T cal. mol-1.K-1 cp(Ti, B) = 6.91 cal. mol-1.K-1 cp(Ti02, s) = 18 + 0.28 x 10-37 - 4.35 x 1057-2 cal. mol-1.K-1 cp(02, g) = 7.16 + 1.0 x 10-3T - 0.4 x 1057T-2 cal. mol-1.K-1 Tila) transform to Ti(ß) at 882 °C, with a heat of transformation equal to 830 cal. mol-1. Calculate the enthalpy change when Ti(B) at 1400 °C is oxidized by pure 02(g) isothermally to form Ti02(s) at 1400 °C. (20 %) AHf,298 K o (Ti02) = -225.5 kcal. mol-1 cp(Ti, a) = 5.48 + 2.25 x 10-3T cal. mol-1.K-1 cp(Ti, B) = 6.91 cal. mol-1.K-1 cp(Ti02, s) = 18 + 0.28 x 10-37 - 4.35 x 1057-2 cal. mol-1.K-1 cp(02, g) = 7.16 + 1.0 x 10-3T - 0.4 x 1057T-2 cal. mol-1.K-1
Expert Answer:
Answer rating: 100% (QA)
In this case first we have the conversion Tialpha Tibeta We have 1155 cal 830 mol... View the full answer

Posted Date:
Students also viewed these accounting questions
-
Calculate the enthalpy change for the reaction P4O6 + 2 O2(g) P4O10(s) given the following enthalpies of reaction: Pals) + 302(g) _ P406(s) P4(s) + 5 Odg)- P4010(s) --1 640.1 kJ --2940.1 kJ
-
The enthalpy change when liquid methanol, CH3OH, vaporizes at 25oC is 38.0 kJ/mol. What is the entropy change when 1.00 mol of vapor in equilibrium with liquid condenses to liquid at 25oC? The...
-
Calculate the enthalpy change for the following reaction: 3NO2(g) + H2O(l) 2HNO3(aq) + NO(g) Use standard enthalpies of formation.
-
Exhibits 7.17 and 7.18 provide footnote excerpts to the financial reports of The Coca-Cola Company and Eli Lilly and Company that discuss the stock option grants given to the employees of the two...
-
What is reasonable assurance? Is it good?
-
A commonly used numerical operation called the first backward difference is defined as y[n] = (x[n]) = x[n] x[n 1], where x[n] is the input and y[n] is the output of the first-backward-difference...
-
A batch total that is calculated by summing the part numbers sold in a batch of 50 sales invoices is called a a. financial total c. record count b. hash total d. part count
-
Assume that you recently accepted a position with Stockman National Bank as an assistant loan officer. As one of your first duties, you have been assigned the responsibility of evaluating a loan...
-
"Competition is at the heart of capitalism. Even if you think there are better ways to set up an economy, you can't deny that competition motivates people to work harder. It forces businesses to...
-
What similarities exist with the job activities of the owner of an automotive repair shop that employs two people and the executive director of the Canadian Cancer Society?
-
How does chronic stress influence physiological systems such as the endocrine and cardiovascular systems, and what are the long-term implications for overall health ? Explain
-
A 4.3 kg cannon ball is flying at 17.3 m/s [E] when it explodes into two fragments. One 1.5 kg fragment (A) goes off at 7.1 m/s [56 o S of E]. What will be the magnitude of the velocity of the second...
-
A large steel ship is partially submerged and motionless in a still sea ("floating"). The steel has a very high density, but the air displaced by the boat has very low density. Together, the average...
-
A cat chases a mouse across a 1.0 m high table. The mouse steps out of the way, and the cat slides off the table and strikes the floor 2.2 m away from the edge of the table the weight of the cat is...
-
Mickley Company's plantwide predetermined overhead rate is $21.00 per direct labor-hour and its direct labor wage rate is $13.00 per hour. The following information pertains to Job A-500: Direct...
-
One strategy in a snowball fight is to throw a snowball at a high angle over level ground. While your opponent is watching the first one, a second snowball is thrown at a low angle timed to arrive...
-
f ( x ) = 1 0 e ^ x + x ^ 6
-
Flicker, Inc., a closely held corporation, acquired a passive activity this year. Gross income from operations of the activity was $160,000. Operating expenses, not including depreciation, were...
-
The following data (and annotations) are related to the June 2019 charges appearing in the work-in-process account for Sutter Company's first processing department: Sutter uses the FIFO method....
-
Terrace Corporation makes an industrial cleaner in two sequential departments, Compounding and Drying. All materials are added at the beginning of the process in the Compounding Department....
-
The following are selected operating data for Jackson Company's Blending Department for November 2019. Painting and packaging operations are carried out subsequently in other departments. Calculate...

Study smarter with the SolutionInn App

