To defend blood volume against dehydration, the body O releases antidiuretic hormone (ADH). O releases atrial...
Fantastic news! We've Found the answer you've been seeking!
Question:

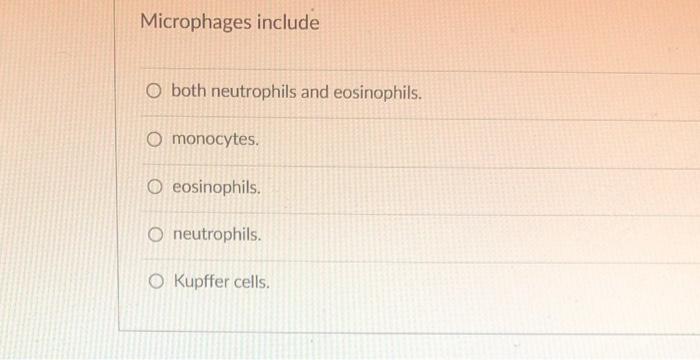


Transcribed Image Text:
To defend blood volume against dehydration, the body O releases antidiuretic hormone (ADH). O releases atrial natriuretic peptide. O reduces reabsorption of water at the kidneys. O experiences a decrease in the blood colloidal osmotic pressure. experiences an increase of interstitial fluids. Microphages include O both neutrophils and eosinophils. O monocytes. O eosinophils. O neutrophils. O Kupffer cells. A crime scene investigator found that a biological fluid sample contains a large amount of IgA-type antibody. This fluid is probably O serum. tears. O blood. O lymph. O interstitial, Hormones of the immune system include all of the following except O tumor necrosis factor. O natriuretic factor. O interferons. O thymosins. O interleukins. To defend blood volume against dehydration, the body O releases antidiuretic hormone (ADH). O releases atrial natriuretic peptide. O reduces reabsorption of water at the kidneys. O experiences a decrease in the blood colloidal osmotic pressure. experiences an increase of interstitial fluids. Microphages include O both neutrophils and eosinophils. O monocytes. O eosinophils. O neutrophils. O Kupffer cells. A crime scene investigator found that a biological fluid sample contains a large amount of IgA-type antibody. This fluid is probably O serum. tears. O blood. O lymph. O interstitial, Hormones of the immune system include all of the following except O tumor necrosis factor. O natriuretic factor. O interferons. O thymosins. O interleukins. To defend blood volume against dehydration, the body O releases antidiuretic hormone (ADH). O releases atrial natriuretic peptide. O reduces reabsorption of water at the kidneys. O experiences a decrease in the blood colloidal osmotic pressure. experiences an increase of interstitial fluids. Microphages include O both neutrophils and eosinophils. O monocytes. O eosinophils. O neutrophils. O Kupffer cells. A crime scene investigator found that a biological fluid sample contains a large amount of IgA-type antibody. This fluid is probably O serum. tears. O blood. O lymph. O interstitial, Hormones of the immune system include all of the following except O tumor necrosis factor. O natriuretic factor. O interferons. O thymosins. O interleukins. To defend blood volume against dehydration, the body O releases antidiuretic hormone (ADH). O releases atrial natriuretic peptide. O reduces reabsorption of water at the kidneys. O experiences a decrease in the blood colloidal osmotic pressure. experiences an increase of interstitial fluids. Microphages include O both neutrophils and eosinophils. O monocytes. O eosinophils. O neutrophils. O Kupffer cells. A crime scene investigator found that a biological fluid sample contains a large amount of IgA-type antibody. This fluid is probably O serum. tears. O blood. O lymph. O interstitial, Hormones of the immune system include all of the following except O tumor necrosis factor. O natriuretic factor. O interferons. O thymosins. O interleukins.
Expert Answer:
Answer rating: 100% (QA)
When body is dehydrated and blood volume is low it secrets Antidiuretic hormone It secrets from the ... View the full answer

Related Book For 

Biochemistry Concepts and Connections
ISBN: 978-0321839923
1st edition
Authors: Dean R. Appling, Spencer J. Anthony-Cahill, Christopher K. Mathews
Posted Date:
Students also viewed these accounting questions
-
An object whose weight 100 lbf experiences a decrease in kinetic energy of 500 ft. lbf and an increase in potential energy of 1500 ft.lbf. The initial velocity and elevation of the object, each...
-
If a company experiences a decrease in its CM ratio, what will be the impact on its break-even level of sales?
-
The osmotic pressure of blood at 37C is 7.7 atm. A solution that is given intravenously must have the same osmotic pressure as the blood. What should be the molarity of a glucose solution to give an...
-
In a bank reconciliation, deposits in transit are: (a) deducted from the book balance. (b) added to the book balance. (c) added to the bank balance. (d) deducted from the bank balance.
-
Tammy, a resident of Virginia, is considering whether to purchase a North Carolina bond that yields 4.6% before tax. She is in the 35% Federal marginal tax bracket and the 5% state marginal tax...
-
Hasbro and Mattel, Inc., are the two largest toy companies in North America. Condensed liabilities and stockholders' equity from a recent balance sheet are shown for each company as follows (in...
-
In Problem 2.4 you were asked to compute a $95 %$ CI on mean gasoline prediction interval on mileage when the engine displacement $x_{1}=275$ in. $^{3}$ Compare the lengths of these intervals to the...
-
Often research and development costs provide companies with benefits that last a number of years. (For example, these costs can lead to the development of a patent that will increase the companys...
-
On December 31, 2023, Dow Steel Corporation had 600,000 shares of common stock and 300,000 shares of 8%, noncumulative, nonconvertible preferred stock issued and outstanding. Dow issued a 4% common...
-
Start with the partial model in the file Ch21 P08 Build a Model.xlsx on the textbook's Web site. Kasperov Corporation has an unlevered cost of equity of 12% and is taxed at a 40% rate. The 4-year...
-
TRUE FALSE 1. Common examples of sin taxes include the taxes imposed on airline tickets and gA sales tax is a common example of a progressive tax rate structure 2. In addition to raising revenues,...
-
When evaluating qualified plans, it is important to remember that pension plans may best suit employers with unpredictable cash flows due to their discretionary funding feature. When evaluating...
-
When has Equity Theory caused you to lower your efforts in the workplace? Explain how this happened referring to the theory?
-
Rewrite this, but keep the quotes the same: It's noted that, "75% of accounting students believe that the AICPA Student Affiliate membership is helpful for their professional goals," indicating the...
-
Acme, Inc. instructed its bank, AJM Bank, to pay $500,000 to TreeTop, Inc. TreeTop, Inc. was also a customer of AJM Bank. AJM Bank executed the payment order by crediting Treetop's account with...
-
1. LPs contribute 90% of the equity and fund 90% of any subsequent investments. 2. GPs contribute 10% of the equity and fund 10% of any subsequent investments. 3. All partners earn an 11% preferred...
-
Given the following class, how would you make it immutable, Re-write it by making the necessary changes. public class Movie ( (b) } public String title; public String director; public int runTime;...
-
Research corporate acquisitions using Web resources and then answer the following questions: Why do firms purchase other corporations? Do firms pay too much for the acquired corporation? Why do so...
-
Give structures for the following, based on the data in Table 10.1. In Table 10.1 a. cis-9-Dodecenoic acid b. 18:1c 11 c. A saturated fatty acid that should melt below 30 °C TABLE 10.1 Some...
-
DNA polymerase requires both a template, to be copied, and a primer, which provides a 3² hydroxyl from which polymerase can extend. Yet this molecule supports DNA polymerase activity. Explain....
-
Calculate the pH values and draw the titration curve for the titration of 500 mL of 0.010 M acetic acid (pKa 4.76) with 0.010 M KOH.
-
A study of photosynthesis in phytoplankton in the open ocean used short pulses of laser light to trigger photosynthetic reactions. The investigators system used 0.10 mW pulses of 640 nm laser light...
-
One molecule of ATP provides 0.30 eV when it is used to power cellular processes. Photosynthesis in a typical plant requires 8 photons at 550 nm to produce 1 molecule of ATP. What is the overall...
-
Osries support for the current tax year came from the following sources: Did Osrie provide more than 50% of his own support? Osrie's wages $9,000 (Osrie spent $6,000 of these funds.) Osrie's mother...

Study smarter with the SolutionInn App


