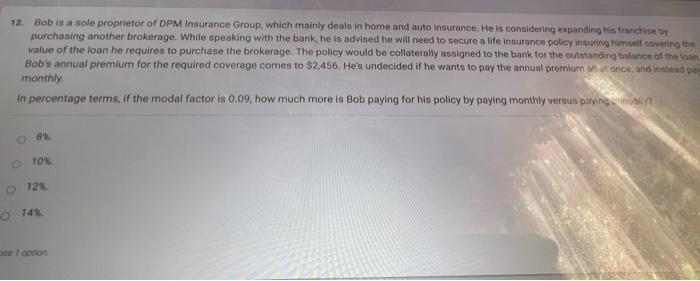
12. Bob is a sole proprietor of DPM Insurance Group, which mainly deals in home and...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
12. Bob is a sole proprietor of DPM Insurance Group, which mainly deals in home and auto insurance. He is considering expanding his tranchise by purchasing another brokerage. While speaking with the bank, he is advised he will need to secure a life insurance policy insuring himself covering the value of the loan he requires to purchase the brokerage. The policy would be collaterally assigned to the bank for the outstanding balance of the loan Bob's annual premium for the required coverage comes to $2,456. He's undecided if he wants to pay the annual premium all at once, and instead play monthly. in percentage terms, if the modal factor is 0.09, how much more is Bob paying for his policy by paying monthly versus paying only 81 O 10% O 12% O 14% se 7 option 12. Bob is a sole proprietor of DPM Insurance Group, which mainly deals in home and auto insurance. He is considering expanding his tranchise by purchasing another brokerage. While speaking with the bank, he is advised he will need to secure a life insurance policy insuring himself covering the value of the loan he requires to purchase the brokerage. The policy would be collaterally assigned to the bank for the outstanding balance of the loan Bob's annual premium for the required coverage comes to $2,456. He's undecided if he wants to pay the annual premium all at once, and instead play monthly. in percentage terms, if the modal factor is 0.09, how much more is Bob paying for his policy by paying monthly versus paying only 81 O 10% O 12% O 14% se 7 option
Expert Answer:
Answer rating: 100% (QA)
Step 11 First we need to find the denominator to divide the annual premium with T... View the full answer

Related Book For 

Fundamental Accounting Principles Volume II
ISBN: 978-1260305838
16th Canadian edition
Authors: Kermit Larson, Tilly Jensen, Heidi Dieckmann
Posted Date:
Students also viewed these accounting questions
-
Located on the outskirts of a large city, the suburb of Peaceful Valley comprises approximately 6,000 upscale homes. The subdivision came about 10 years ago when a developer built an earthen dam on...
-
Levi Strauss has some of its jeans stone-washed under a contract with independent U.S. Garment Corp. U.S. Garment's operating cost per machine is $22,000 for year 1 and then it increases by $1000 per...
-
A hundred or so boats go fishing every year for three or four weeks off of the Bering Strait for Alaskan king crabs. To catch these king crabs, large pots are baited and left on the sea bottom often...
-
Pam has a $25,900 basis (including her share of debt) in her 50 percent partnership interest in the Meddoc Partnership before receiving any distributions. This year Meddoc makes a current...
-
A sample of 200 teenagers shows that 50 believe that war is inevitable, and a sample of 300 people over age 60 shows that 93 believe war is inevitable. Is the proportion of teenagers who believe war...
-
One mole of an ideal gas is contained in a cylinder with a movable piston. The initial pressure, volume, and temperature are Pi, Vi, and Ti, respectively. Find the work done on the gas for the...
-
The revenue recognition principle says a. record revenue only after you have earned it. b. record revenue only when you receive cash. c. match revenues and expenses in order to compute net income. d....
-
Calculating the materials usage variance Laura Pierre is the manager of the Roebuck Bagel Shop. The corporate office had budgeted her store to sell 3,000 ham sandwiches during the week beginning July...
-
How do leadership styles, such as transformational leadership, shared leadership, and servant leadership, impact team dynamics, member motivation, and overall team effectiveness ?
-
Insert the following data into your database separating it appropriately into the make and model tables and setting the make_id foreign key to link each model to its corresponding make. make model...
-
A patient has developed a fluid imbalance secondary to MDMA use which has resulted in a condition called Syndrome of Inappropriate Antidiuretic Hormone Secretion (SIADH) 1) On A concept map, start at...
-
What is whirling?
-
A simple pendulum of length \(0.8 \mathrm{~m}\) is released from the initial position of \(80^{\circ}\) from the vertical. How long does it take to reach the position \(\theta=0^{\circ}\) ?
-
How is the critical speed of a shaft determined?
-
Fill in the Blank. The vertical component of the inertia force of a reciprocating engine has primary and ____________ parts.
-
True or False. The oil whip in the bearings can cause instability in a rotor system.
-
You are taking an eye exam. You stand 5 m from a board that has letters printed on it. The separation between two of the letters on the board is 1 cm. Assume that the light in the room has a...
-
Refer to the data for problem 13-36 regarding Long Beach Pharmaceutical Company. Required: Compute each division's residual income for the year under each of the following assumptions about the...
-
Calculate the return on common shareholders? equity for 2020 and evaluate the result against the industry average in Exhibit 17.11, explaining why it compares favourably or unfavourably (round to two...
-
On October 3, 2020, Synthetic Inc, a cell phone protector company, issued 4,000 of its preferred shares for cash of $15 each. On November 19 the company issued 3,400 preferred shares in exchange for...
-
Refer to the information in Problem 16-5B. Prepare a statement of cash flows for 2020 using the direct method to report cash inflows and outflows from operating activities. Other information: a. All...
-
When there is a concentration gradient in the system, show that the potential gradient is composed of two terms, (i) an Ohm's-law contribution and (ii) a diffusional contribution. State the equation...
-
Copper is deposited at a cathode from solution with a bulk concentration of \(0.5 \mathrm{M}\) at the rate of \(3.0 \mathrm{~g} / \mathrm{m}^{2} \cdot \mathrm{s}\). Find the surface concentration of...
-
Find the mobility of \(\mathrm{H}^{+}, \mathrm{OH}^{-}\), and other ions from the diffusivity data given in Table 22.1. Table 22.1. Diffusion coefficients of ions in water at 25 C. H+ Na+ K+ Ca+ OH-...

Study smarter with the SolutionInn App


