Question: Use the Lewis structure for the amine below. Match the Lewis structure to the correct condensed structure and the correct skeletal structure. Lewis structure:
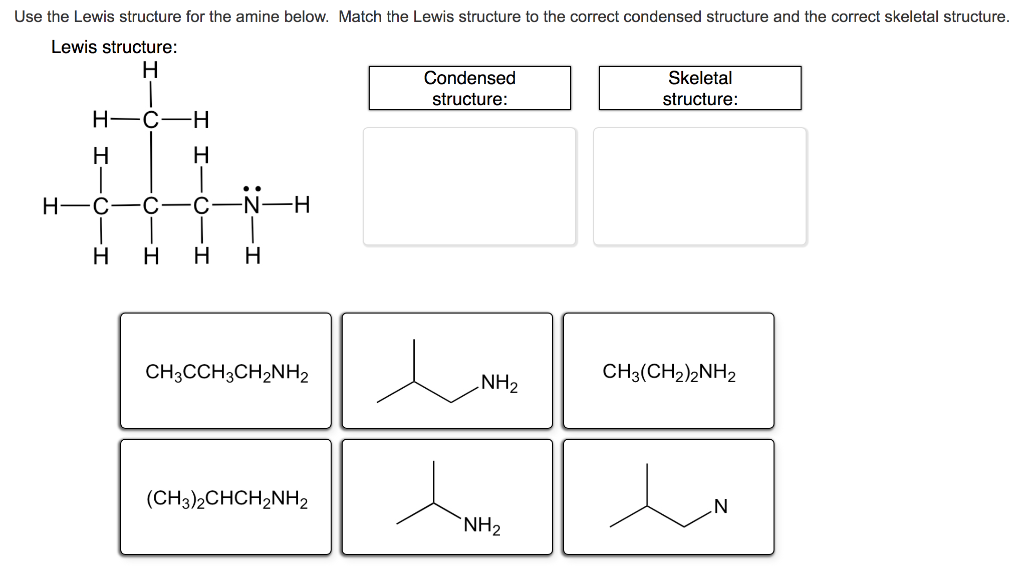
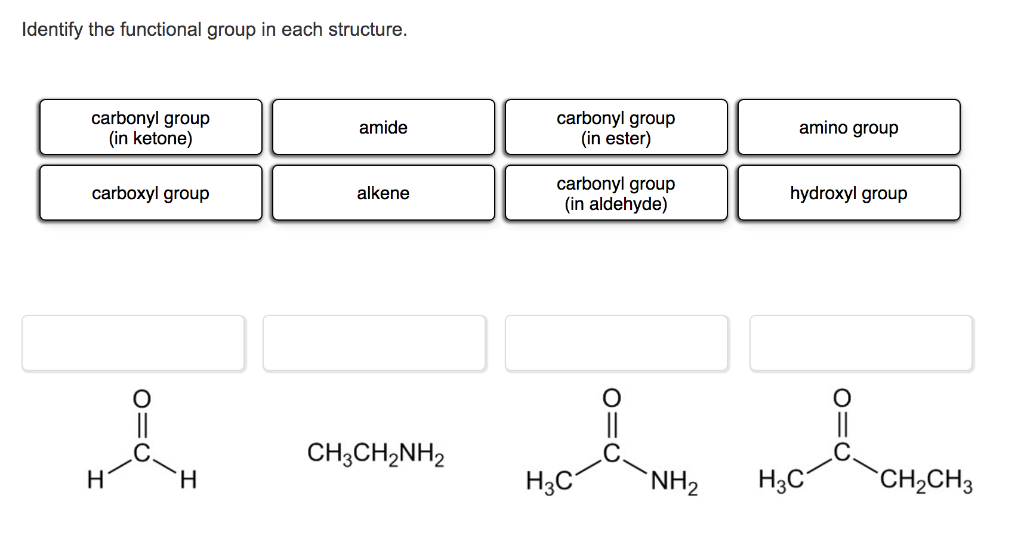

Use the Lewis structure for the amine below. Match the Lewis structure to the correct condensed structure and the correct skeletal structure. Lewis structure: H Condensed Skeletal structure: structure: H-C -H- H H .. H-C C-N-H H H H H CH3CCH3CH2NH2 CH3(CH2)2NH2 NH2 (CH3)2CHCH2NH2 `NH2 N Identify the functional group in each structure. carbonyl group (in ketone) carbonyl group (in ester) amide amino group carbonyl group (in aldehyde) carboxyl group alkene hydroxyl group CH3CH,NH2 H H. H3C `NH2 H3C `CH2CH3 Which one of the following structures represents an unstable organic compound that is not likely to exist? H H. H H. H. H- C- C-C H H. CH2=CHCH2CH3 CH3CH2CH(CH3)2
Step by Step Solution
3.40 Rating (166 Votes )
There are 3 Steps involved in it
Lewis structure H HCH H HCCCNH HHHH carbonyl group in aldehyde 0 O H H H H OH amino group CCCH H ... View full answer

Get step-by-step solutions from verified subject matter experts


