Question: (Use the pull-down boxes to specify states such us (ag) or (s). If a box is not needed, leave it blank. If no reaction

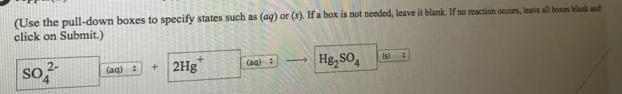
(Use the pull-down boxes to specify states such us (ag) or (s). If a box is not needed, leave it blank. If no reaction occuns, leave all bones blank nd click on Submit.) 2Ag Co 2- (aq) : As, Co (Use the pull-down boxes to specify states such as (ag) or (3). If a box is not needed, leave it blank. If no reaction occuns, leave all bons blank and click on Submit.) 2- SO 2Hg Hg, so, (ag) : (ag) : Is)
Step by Step Solution
There are 3 Steps involved in it
Reaction and hydiochyic acid eg combined OC Cur when Mnls are Mm s Reactivity series in ... View full answer

Get step-by-step solutions from verified subject matter experts


