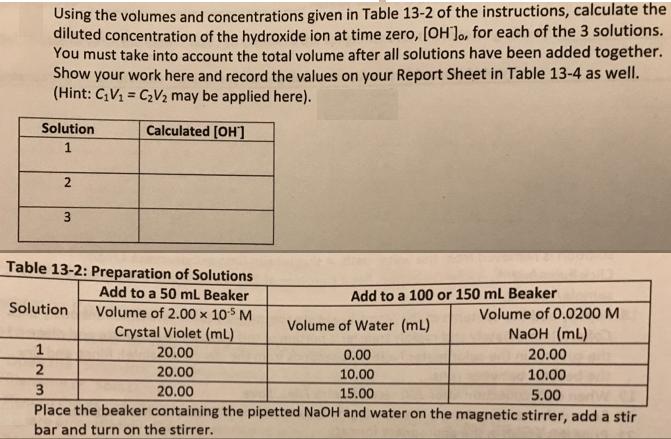
Using the volumes and concentrations given in Table 13-2 of the instructions, calculate the diluted concentration...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Using the volumes and concentrations given in Table 13-2 of the instructions, calculate the diluted concentration of the hydroxide ion at time zero, [OH]o, for each of the 3 solutions. You must take into account the total volume after all solutions have been added together. Show your work here and record the values on your Report Sheet in Table 13-4 as well. (Hint: CV = C2V2 may be applied here). Solution Calculated [OH'] 1. Table 13-2: Preparation of Solutions Add to a 50 mL Beaker Add to a 100 or 150 mL Beaker Solution Volume of 2.00 x 105 M Crystal Violet (mL) 20.00 Volume of 0.0200 M Volume of Water (mL) NaOH (mL) 20.00 0.00 20.00 10.00 10.00 3 20.00 15.00 5.00 Place the beaker containing the pipetted NaOH and water on the magnetic stirrer, add a stir bar and turn on the stirrer. Using the volumes and concentrations given in Table 13-2 of the instructions, calculate the diluted concentration of the hydroxide ion at time zero, [OH]o, for each of the 3 solutions. You must take into account the total volume after all solutions have been added together. Show your work here and record the values on your Report Sheet in Table 13-4 as well. (Hint: CV = C2V2 may be applied here). Solution Calculated [OH'] 1. Table 13-2: Preparation of Solutions Add to a 50 mL Beaker Add to a 100 or 150 mL Beaker Solution Volume of 2.00 x 105 M Crystal Violet (mL) 20.00 Volume of 0.0200 M Volume of Water (mL) NaOH (mL) 20.00 0.00 20.00 10.00 10.00 3 20.00 15.00 5.00 Place the beaker containing the pipetted NaOH and water on the magnetic stirrer, add a stir bar and turn on the stirrer.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What are the considerations/particularities you must take into account when. Motivating Negotiating In a diverse workplace environment different from a non-diverse environment?
-
You add 1.5 mL of 1 M HCl to each of the following solutions. Which one will show the least change of pH? a. 15 mL of 0.1 M NaOH b. 15 mL of 0.1 M HC2H3O2 c. 30 mL of 0.1 M NaOH and 30 mL of 0.1 M...
-
Calculate the volume in mL of a 1.420 M NaOH solution required to titrate the following solutions: (a) 25.00 mL of a 2.430 M HCl solution (b) 25.00 mL of a 4.500 M H2 SO4 solution (c) 25.00 mL of a...
-
Monroe Inc. is an all-equity firm with 500,000 shares outstanding. It has $2,000,000 of EBIT, and EBIT is expected to remain constant in the future. The company pays out all of its earnings, so...
-
Discuss the major principle that describes recording expenses.
-
1. What type(s) of power does Frank Stronach appear to possess? 2. Can Stronach be described as a visionary leader? If so, why? 3. Which of the classic leadership styles does Stronach appear to...
-
What stakeholder affected by their behavior was Gilead weighting very lightly when it decided what to tell the FDA about the medicines it wished to have permission to sell?
-
Your systems analysis team is close to completing a system for Meecham Feeds. Roger is quite confident that the programs that he has written for Meechams inventory system will perform as necessary,...
-
Te If you have $90,000 in a bank account that is paying an interest rate of 6 percent that is being compounded annually, how many years will it take to double your investment if the interest rate...
-
Using exponential smoothing, calculate the forecasts for the same months as in problem 8.3c. The old average for month 3 was 96 and = 0.4. What is the difference between the two forecasts for month...
-
Let G = (V,E) be a simple graph with |V| 2. The complement graph G of G is the simple graph whose vertex set is V and whose edge set consists of all the edges that have as endpoints nonadjacent...
-
The original mortgage balance (MB0) is $350,000; and, the number of months of the mortgage loan (n) is 360. What is the monthly mortgage payment (MP) if the initial note rate (nominal annual rate)...
-
The market study carried out by Turki indicates that the cost of building a date factory in Virginia, USA costs KD 250,000. The additional working capital is estimated to be KD 60,000. Turki intends...
-
What are the most important things you do as a financial risk manager to ensure your bank's financial health? "Liquidity is a Bank's Capacity to fund increase in assets and meet both Expected and...
-
Describe one example of organizational citizenship behavior that you have observed someone demonstrated at work or yourself have demonstrated at work. What are the major reasons for this person to...
-
How much is assumed collectible from the debtor How much should be included as part of the gross estate of Mr. San Diego. 1. Mr. San Diego's debtor suffered extreme financial setback that he as frank...
-
Starting from rest, a 920 kg car coasts a distance of 8.00m downhill along a road that makes an angle of 30.0 below the horizontal. There is no frictional force that acts on the car. The car's...
-
Two mutually exclusive investment alternatives are being considered. Alternative A requires an initial investment of $20,000 in a machine. Annual operating and maintenance costs are anticipated to be...
-
Three apprentice tailors (X, Y, and Z) are assigned the task of measuring the seam of a pair of trousers. Each one makes three measurements. The results in inches are X (31.5, 31.6, 31.4); Y (32.8,...
-
Match each of the following energy changes with one of the processes given: ionization energy, electron affinity, bond enthalpy, and standard enthalpy of formation. (a) F(g) + e - F- (g) (b) F2(g) ...
-
Referring to the metabolic process involving glucose on p. 826, calculate the maximum number of moles of ATP that can be synthesized from ADP from the breakdown of one mole of glucose.
-
Should morality, in and of itself, be considered a sufficient basis for defining particular conduct as criminal?
-
What are the chief distinctions between the civil and criminal law? Why do the criminal and civil law sometimes overlap?
-
What means of punishment for criminal offenses exist in your state? Is capital punishment available for persons convicted of first-degree murder? Which punishments, if any, do you think are most...

Study smarter with the SolutionInn App



