VIRTUAL AIR (PTY LTD is a smal company which, bncause of its aggresive adverteing campaigna, has...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
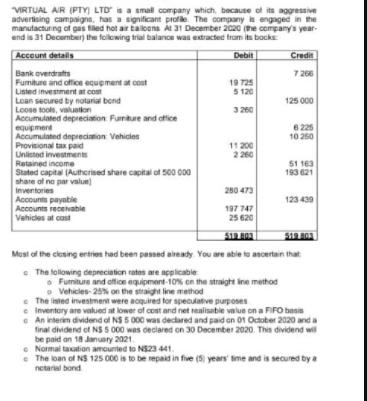
VIRTUAL AIR (PTY LTD is a smal company which, bncause of its aggresive adverteing campaigna, has a significant profie The company in engaged in the manutacturing of gas tiled hot air tailoana At 31 December 2020 (the company's year- end is 31 December) the folowing trial balance was extracted tram its bocks Acceunt details Debit Credit 7 266 Bank overdrafts Fumiture and office equement at cost Listed investmernt at com Loan secured by notarial bond Loose toole, valuatkon Accumulated depreciation Furnture and ofice equipment Accumulated depreciation Vehicies Provisional tax paid Unistod investments Retained income Stated capital (Autherised share capital of 500 000 shate of no par valun) Inventories Accounts payatile Accounts recevabie Vehices at cost 19 725 5 120 3 260 125 000 6 225 10 260 11 200 2 260 S1 163 193 21 280 473 123 439 197 747 25 620 Must of the clesing ertrien had been pessed aireaty You are able to ancertan that e The tollowing depreciation rates are applicabie • Fumiture and ofioe equipment-10% cn the straight ine method o Vehicles- 25% on the straight line method e The isted investment were acquired for speculative purposes e Inventory are valued at lower of cost and net realisable value on a FIFO basis e An interim dividend of NS 5 000 was decared and paid an O1 October 2020 and a final dividend ef NS 5 000 was declared on 30 December 2020. This dividend wil be paid an 18 January 2021 e Normal taation amounted to NS23 441. e The loan of NS 125 000 is to be repaid in five (5 years' ime and is secured by a netarial bond T A TO 31 ete are retee of HSn i A 31 bete aN Eme FRECoie su 5 urta 21 etn 24 VIRTUAL AIR (PTY LTD is a smal company which, bncause of its aggresive adverteing campaigna, has a significant profie The company in engaged in the manutacturing of gas tiled hot air tailoana At 31 December 2020 (the company's year- end is 31 December) the folowing trial balance was extracted tram its bocks Acceunt details Debit Credit 7 266 Bank overdrafts Fumiture and office equement at cost Listed investmernt at com Loan secured by notarial bond Loose toole, valuatkon Accumulated depreciation Furnture and ofice equipment Accumulated depreciation Vehicies Provisional tax paid Unistod investments Retained income Stated capital (Autherised share capital of 500 000 shate of no par valun) Inventories Accounts payatile Accounts recevabie Vehices at cost 19 725 5 120 3 260 125 000 6 225 10 260 11 200 2 260 S1 163 193 21 280 473 123 439 197 747 25 620 Must of the clesing ertrien had been pessed aireaty You are able to ancertan that e The tollowing depreciation rates are applicabie • Fumiture and ofioe equipment-10% cn the straight ine method o Vehicles- 25% on the straight line method e The isted investment were acquired for speculative purposes e Inventory are valued at lower of cost and net realisable value on a FIFO basis e An interim dividend of NS 5 000 was decared and paid an O1 October 2020 and a final dividend ef NS 5 000 was declared on 30 December 2020. This dividend wil be paid an 18 January 2021 e Normal taation amounted to NS23 441. e The loan of NS 125 000 is to be repaid in five (5 years' ime and is secured by a netarial bond T A TO 31 ete are retee of HSn i A 31 bete aN Eme FRECoie su 5 urta 21 etn 24
Expert Answer:
Answer rating: 100% (QA)
ANSWER 1 Calculations Note 1 Taxation Taxation 23441 Less Tax Provisions 11200 Closing Provision 12241 Note2 Retained Income Opening Balance 51163 Les... View the full answer

Related Book For 

Financial Accounting and Reporting
ISBN: 978-0273744443
14th Edition
Authors: Barry Elliott, Jamie Elliott
Posted Date:
Students also viewed these accounting questions
-
You are able to buy an investment today for $1,000 that gives you the right to receive $438 in each of the next three years. What is the internal rate of return on this investment?
-
Kids Sports Consulting Pty Ltd is a company set up by sports and recreation management students to gain experience in running their own business. It had the following contribution margin income...
-
Solomon Pty Ltd is a highly successful, closely held Brisbane company that manufactures and assembles motor vehicle speciality parts that are sold in auto parts stores. Sales and profits have...
-
Fletcher, Inc. disposes of under- or over-applied overhead at year-end as an adjustment to cost of goods sold. Prior to disposal, the firm reported cost of goods sold of $619,000 in a year when...
-
How does HIV confound the human immune system?
-
Harrow Seed and Fertilizer compiled the following estimates for its operations. Capacity per period is sales of $150 000. For each of the following, perform a break-even analysis showing (a) An...
-
Derringer and Suich ("Simultaneous Optimization of Several Response Variables," Journal of Quality Technology, 1980) studied the relationship of an abrasion index for a tire tread compound in terms...
-
Miranda, Inc., manufactures and sells commercial and residential security equipment. The comparative unclassified balance sheets for December 31, 2011 and 2010 are provided below. Selected missing...
-
Image transcription text Page AND To Trojedt 2.2 Maintenance Record Entries Name: Class Section: Aeronautical Product Model / Registration Number Serial Number (S/N) ITIS Aircraft - mig 1983 Cessna...
-
What are the four kinds of resources?
-
4. Translate the following C code to MIPS assembly (in two separate files). Run the program step by step and observe the order of instructions being executed and the value of $sp. int main() { int...
-
Would a polar protic solvent like ethanol favor the solvolysis of 2-chloro-2-methylpropane? Why or why not? View Available Hint(s) O Ethanol would favor the solvolysis; the mechanism is E1. O Ethanol...
-
Prepare a 0.15% (w/v) solution of haemoglobin in 10 mM HCl pH2. Dilute to 0.03, 0.05, 0.07% and 0.09% in 10 mM HCl pH 2. 2. Prepare a calibration curve of haemoglobin in 10mM HCl pH 2 at 372 nm...
-
What happens to the pressure of a sample of gas if it is allowed to exp three times its original volume? The temperature remains constant. Pressure is reduced by a third Pressure is halved Pressure...
-
3. Benzoic acid + methanol III. Write the equation for the acid hydrolysis of the following esters: 0 11 1. H -C-0-CHz - CH3 2. Ethyl butanoate heat 3. Methyl propanoate Page
-
An experimental procedure for standardization of the titrant states the following: Weigh accurately, about 0.26-0.27 g of the primary-standard. Record the mass to the nearest 0.0001 g. Which mass...
-
As shown below, particle C is located at (0 cm, 16 cm), particle D is at the origin, & particle E is at (16 cm, 0 cm). Additionally, ac = 4 C, qp = -2 C, & q = 3 C. D E A.) Determine the magnitude &...
-
Compare and contrast debt financing and equity financing as ways of starting a new business. Does one have an overall advantage over the other? What situation is more favorable to the use of debt...
-
Charles plc is applying IAS 32 and IAS 39 for the first time this year and is uncertain about the application of the standard. Charles plc balance sheet is as follows: Note 1. The forward contracts...
-
The major shareholder/director of Esrever Ltd has obtained average data for the industry as a whole. He wishes to see what the forecast results and position of Esrever Ltd would be if in the ensuing...
-
At 1 January 20X5 Bridge Finance plc agreed to finance the lease of machinery costing $37,200 to Rapid Growth plc at a lease cost of $10,000 per annum payable at the end of the year, namely 31...
-
While the BohrSommerfeld condition sometimes gets the energy eigenvalues exactly correct, it can also be used for systems where the exact solution is not known. In this example, we will estimate the...
-
While we introduced the variational method and the power method both as a way to approximate the ground state of some system, they both can be used to approximate excited states as well, with...
-
It's useful to see how our quantum perturbation theory works in a case that we can solve exactly. Let's consider a two-state system in which the Hamiltonian is...

Study smarter with the SolutionInn App


