Volumetric analysis of natural gas shows the following: 70% CH4, 10% H2, 15% N2, 2% O2...
Fantastic news! We've Found the answer you've been seeking!
Question:
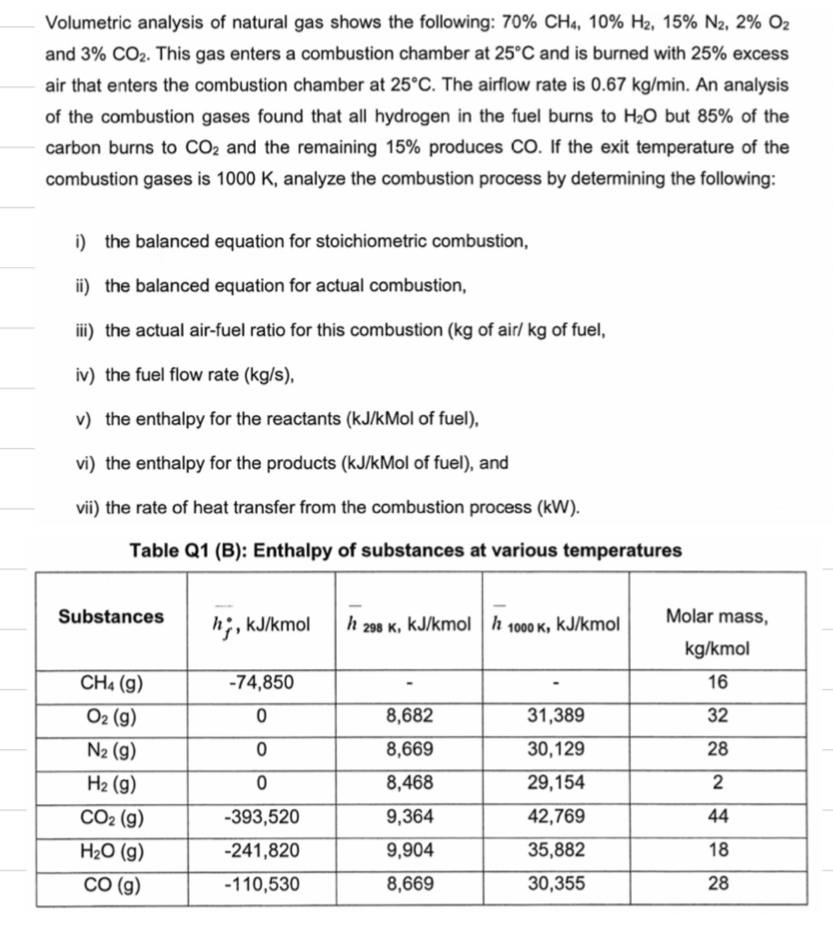
Transcribed Image Text:
Volumetric analysis of natural gas shows the following: 70% CH4, 10% H2, 15% N2, 2% O2 and 3% CO2. This gas enters a combustion chamber at 25C and is burned with 25% excess air that enters the combustion chamber at 25C. The airflow rate is 0.67 kg/min. An analysis of the combustion gases found that all hydrogen in the fuel burns to H2O but 85% of the carbon burns to CO2 and the remaining 15% produces CO. If the exit temperature of the combustion gases is 1000 K, analyze the combustion process by determining the following: i) the balanced equation for stoichiometric combustion, ii) the balanced equation for actual combustion, iii) the actual air-fuel ratio for this combustion (kg of air/ kg of fuel, iv) the fuel flow rate (kg/s), v) the enthalpy for the reactants (kJ/kMol of fuel), vi) the enthalpy for the products (kJ/kMol of fuel), and vii) the rate of heat transfer from the combustion process (kW). Table Q1 (B): Enthalpy of substances at various temperatures Substances h, kJ/kmol h 298 K, kJ/kmol h 1000 K, kJ/kmol Molar mass, kg/kmol CH4 (g) -74,850 16 O2 (g) 0 8,682 31,389 32 N2 (g) 0 8,669 30,129 28 H2 (g) 0 8,468 29,154 2 CO2 (g) -393,520 9,364 42,769 44 HO (g) -241,820 9,904 35,882 18 CO (g) -110,530 8,669 30,355 28 Volumetric analysis of natural gas shows the following: 70% CH4, 10% H2, 15% N2, 2% O2 and 3% CO2. This gas enters a combustion chamber at 25C and is burned with 25% excess air that enters the combustion chamber at 25C. The airflow rate is 0.67 kg/min. An analysis of the combustion gases found that all hydrogen in the fuel burns to H2O but 85% of the carbon burns to CO2 and the remaining 15% produces CO. If the exit temperature of the combustion gases is 1000 K, analyze the combustion process by determining the following: i) the balanced equation for stoichiometric combustion, ii) the balanced equation for actual combustion, iii) the actual air-fuel ratio for this combustion (kg of air/ kg of fuel, iv) the fuel flow rate (kg/s), v) the enthalpy for the reactants (kJ/kMol of fuel), vi) the enthalpy for the products (kJ/kMol of fuel), and vii) the rate of heat transfer from the combustion process (kW). Table Q1 (B): Enthalpy of substances at various temperatures Substances h, kJ/kmol h 298 K, kJ/kmol h 1000 K, kJ/kmol Molar mass, kg/kmol CH4 (g) -74,850 16 O2 (g) 0 8,682 31,389 32 N2 (g) 0 8,669 30,129 28 H2 (g) 0 8,468 29,154 2 CO2 (g) -393,520 9,364 42,769 44 HO (g) -241,820 9,904 35,882 18 CO (g) -110,530 8,669 30,355 28
Expert Answer:

Posted Date:
Students also viewed these mechanical engineering questions
-
The accountant of your business has recently been taken ill through overwork. In his absence his assistant has prepared some calculations of the profitability of a project, which are to be discussed...
-
THE ETHICAL DIMENSION Is the courts decision in this case fair to employers? Why or why not? Josephine Mora was sixty-two years old when she was fired from her job as a fund-raiser for Jackson...
-
Barkley Corp. obtained a trade name in January 2009, incurring legal costs of $15,000. The company amortizes the trade name over eight years. Barkley successfully defended its trade name in January...
-
Brian Hughes and Wendy Perez formed a partnership five years ago. The partnership has been very successful and is growing rapidly. The partners are evaluating future actions for the next five years....
-
a. Prepare the statement of cash flows, with a total column for the three-year period. (Do not include reconciliation). b. Comment on significant cash flow items in the statement prepared in (a). c....
-
A manager who wants to find the most innovative solution to a problem would benefit from which approach to decision making?
-
Provide an example for each of the five ways a company can gain a competitive advantage through differentiation. In which of these ways does Kraft Heinz gain a competitive advantage? Provide...
-
Byrex has developed a new kind of bypass unit. The sales department forecasts demand of 5,000 units in the first year and a decrease in demand of 10 percent a year after that. After five years, the...
-
A business is thinking about an investment of $28,000,000 (purchase price) in new equipment to replace older equipment with a book value of $12,000,000 and a market value of $20,000,000. If the...
-
CleverTechnology Angel Investor owns 4,000,000 shares (or 16% of shares) of Bluebox Software from a Series A round. The Board of Bluebox Software decides to raise additional $8,500,000 in a Series B...
-
You know that a firm's EBIT(1-t) is $765,000, with CAPEX of $690,000, after-tax interest of $45,000, depreciation of $520,000 and an increase in working capital of $430,000 for the same year. What is...
-
Using mcconnell dowell 2021 annual report. Can u find out what is the a. Assets Ratio of the company and explain what this means. b. Liability Ratio of the company and explain what this means. c....
-
The Roman conquest of Greece swept away the influence of the Greeks, such that Greek culture had little impact on the first-century world. True /False That Peter became the bishop of Rome is found in...
-
A test car is driven a fixed distance of n miles along a straight highway. (Here n Z+.) The car travels at one mile per hour for the first mile, two miles per hour for the second mile, four miles...
-
Describe or define the following terms: 1. Asset 2. Liability 3. Shareholders equity 4. Revenue 5. Expense Discuss how the five terms relate to one another.
-
Discuss the difference between historical and forecasted (or pro forma) financial statements. How are these different sets of financial statements used by managers and shareholders for...
-
Most balance sheets are defined as classified balance sheets in that both the assets and liabilities are dichotomized into the categories of current and noncurrent. [In those industries in which the...

Study smarter with the SolutionInn App

