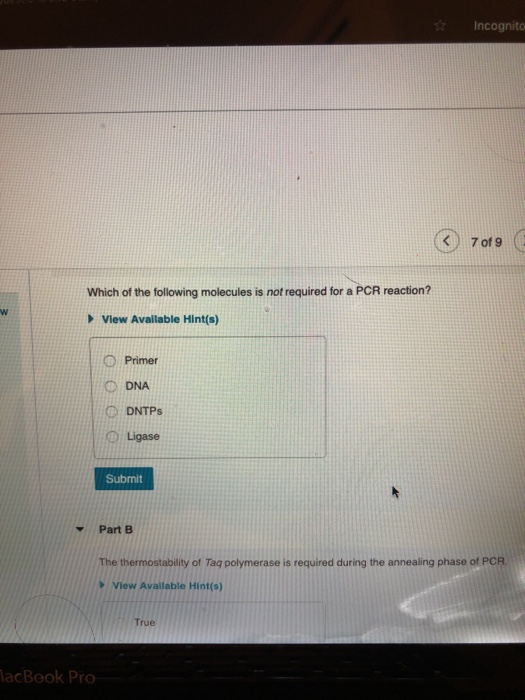
W Y Which of the following molecules is not required for a PCR reaction? View Available...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
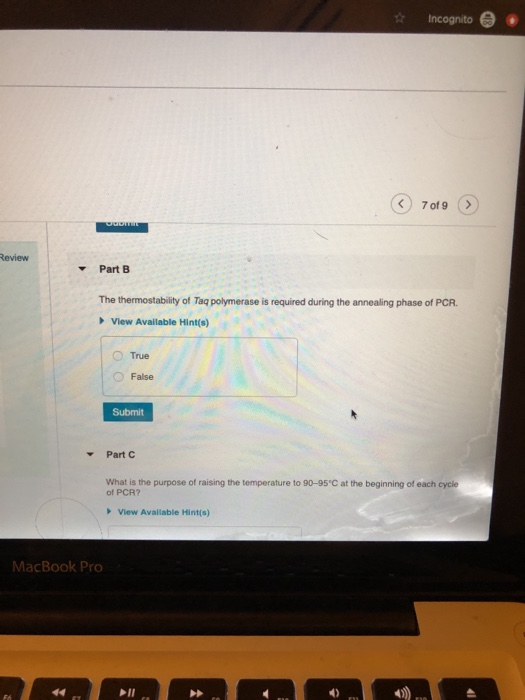
W Y Which of the following molecules is not required for a PCR reaction? View Available Hint(s) MacBook Pro Primer ODNA DNTPs Ligase Submit Part B Incognito True 7 of 9 The thermostability of Taq polymerase is required during the annealing phase of PCR. View Available Hint(s) Review F6 ▾ 44 Odbmit Part B MacBook Pro The thermostability of Taq polymerase is required during the annealing phase of PCR. View Available Hint(s) True False Submit Part C Incognito 7 of 9 View Available Hint(s) What is the purpose of raising the temperature to 90-95°C at the beginning of each cycle of PCR? W Y Which of the following molecules is not required for a PCR reaction? View Available Hint(s) MacBook Pro Primer ODNA DNTPs Ligase Submit Part B Incognito True 7 of 9 The thermostability of Taq polymerase is required during the annealing phase of PCR. View Available Hint(s) Review F6 ▾ 44 Odbmit Part B MacBook Pro The thermostability of Taq polymerase is required during the annealing phase of PCR. View Available Hint(s) True False Submit Part C Incognito 7 of 9 View Available Hint(s) What is the purpose of raising the temperature to 90-95°C at the beginning of each cycle of PCR?
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below Answer D Ligase Ligase is an enzyme tha... View the full answer

Related Book For 

Equity Asset Valuation
ISBN: 978-0470571439
2nd Edition
Authors: Jerald E. Pinto, Elaine Henry, Thomas R. Robinson, John D. Stowe, Abby Cohen
Posted Date:
Students also viewed these accounting questions
-
Which of the following molecules may be polar? (a) CH3Cl (C3J, (b) HW2 (CO)10 (D4h), (c) SnCl4 (Td).
-
Which of the following molecules may show a pure rotational microwave absorption spectrum? (a) H20, (b) H202, (c) NH4 (d) N20?
-
Which of the following molecules may show a pure rotational Raman spectrum? (a) CH2Cl2 (b) CH3CH3, (c) SP6, (d) N2O?
-
Use the data in the table to complete the following. (a) Make a scatterplot of the data. Estimate a value for b so that f(x) = 0.0002x b models the data. (b) Check the accuracy of f(x). (c) The moon...
-
Tropics Fruit Company, based on Oahu, grows, processes, cans, and sells three main pineapple products: sliced, crushed, and juice. T he outside skin is cut off in the Cutting Department and processed...
-
Define ethics and explain what ethical communication encompasses.
-
Consider the oscillation circuit shown in Figure 26.23. During the portion of the cycle when the capacitor's top plate is positive and the amount of charge is increasing, the current through the...
-
Sherman Company employs 400 production, maintenance, and janitorial workers in eight separate departments. In addition to supervising operation, the supervisors of the departments are responsible for...
-
A cost benefit analysis is to be completed on each identified asset. Your analysis would begin with high value asset to lower valued asset. It is important the analysis explains the methods or ways...
-
A physics professor did daredevil stunts in his spare time. His last stunt was an attempt to jump across a river on a motorcycle (Fig. 3.51). The takeoff ramp was inclined at 53.0", the river was...
-
Sweeten Company had no jobs in progress at the beginning of the year and no beginning inventories. It started, completed, and sold only two jobs during the year-Job P and Job Q. The company uses a...
-
You push on a rectangular door at the location of the knob . The door s mass is 6 8 . 4 kg , and its side - to - side width is 1 . 5 4 m . The knob is located 0 . 2 0 m from the right - hand side of...
-
A 5 kg weight is attached to a spring fixed to an inflexible beam. When the weight is at rest, the spring is 1 0 m long. The weight is pulled down from its equilibrium position by 2 m and released....
-
Let s look at the growth of Winston - Salem, NC . In 2 0 2 2 , the population was about 5 3 7 , 0 0 0 . The city s population grew by 9 0 0 0 people from 2 0 2 2 to 2 0 2 3 . a . If the growth...
-
Suppose you place an initial investment in a savings account that offers a 5 % annual interest rate. Calculate the time required for this investment to grow to twice its original amount. Utilize the...
-
A new comet is discovered that orbits the Sun at a distance of 2 8 4 million kilometers. How many astronomical units is that, and about how many days does this asteroid take to go around the Sun...
-
How to write the formulas? + Nested IF + Mixed Ref Given the sales amounts and commission rates, calculate commission amounts, using ONE formula that will be first entered in C3 and then copied to...
-
In the figure, two loudspeakers, separated by a distance of d1 = 2.63 m, are in phase. Assume the amplitudes of the sound from the speakers are approximately the same at the position of a listener,...
-
The management of Telluride, an international diversified conglomerate based in the United States, believes that the recent strong performance of its wholly owned medical supply subsidiary, Sundanci,...
-
Based on the following information, determine whether Vertically Integrated Manufacturing (VIM) earned any residual income for its shareholders: VIM had total assets of $ 3,000,000, financed with...
-
Mohan Gupta is the portfolio manager of an India - based equity fund. He is analyzing the value of Tata Chemicals Ltd. (Bombay Stock Exchange: TATACHEM). Tata Chemicals is Indias leading manufacturer...
-
Depository institutions have been losing their advantage over other financial intermediaries in attracting customers funds. Why?
-
In the aftermath of the financial crisis of 20072009, there were calls to reinstate the separation of commercial and investment banking activities that was removed with the repeal of the...
-
The globalization of banking has led to the need for global benchmarks for interest rates. In light of the LIBOR scandal, what characteristic do you think is most central to any new interest rate...

Study smarter with the SolutionInn App


