We are trying to make 450 kg of 0.15 M NaOH solution by adding ice (-20...
Fantastic news! We've Found the answer you've been seeking!
Question:
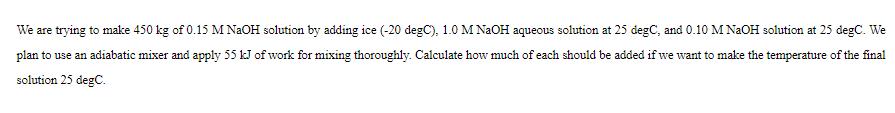
Transcribed Image Text:
We are trying to make 450 kg of 0.15 M NaOH solution by adding ice (-20 degC), 1.0 M NaOH aqueous solution at 25 degC, and 0.10 M NaOH solution at 25 degC. We plan to use an adiabatic mixer and apply 55 kJ of work for mixing thoroughly. Calculate how much of each should be added if we want to make the temperature of the final solution 25 degC. We are trying to make 450 kg of 0.15 M NaOH solution by adding ice (-20 degC), 1.0 M NaOH aqueous solution at 25 degC, and 0.10 M NaOH solution at 25 degC. We plan to use an adiabatic mixer and apply 55 kJ of work for mixing thoroughly. Calculate how much of each should be added if we want to make the temperature of the final solution 25 degC.
Expert Answer:
Answer rating: 100% (QA)
Solution Lets The mass of the 10 M NaOH solution m1 kg The mass of the 010 M NaOH solution m2 kg The energy gained by the final solution is given by E... View the full answer

Related Book For 

Understanding Basic Statistics
ISBN: 9781111827021
6th Edition
Authors: Charles Henry Brase, Corrinne Pellillo Brase
Posted Date:
Students also viewed these accounting questions
-
Case Swimmers Headphones1 Finally! Its the end of 2019 and you are sitting at your new desk in a plush office in north Bogota, with a delicious cup of Colombian coffee. You think about all it took to...
-
Prosci's change management methodology is developed based on research with over 3,400 participants over the last twenty years. What is unique about the methodology is that it comes from real project...
-
The Orange Blossom Marathon takes place in Orlando, Florida, each December. The organizers of this race are trying to solve a problem that occurs at the finish line each year. Thousands of runners...
-
The fraud examiner's report should have what characteristics? a. Accuracy and clarity b. Relevant and material information c. Should not contain opinions or prejudices of fraud examiner. d. All of...
-
If a long rope is hung from a ceiling and waves are sent up the rope from its lower end, they do not ascend with constant speed. Explain.
-
In Figure, length a is 4.7 cm (short) and current i is 13 A. What are the (a) Magnitude and (b) Direction (into or out of the page) of the magnetic field at point P? 2a
-
Assume that Fossil sold watches to a department store on account for \($43,000\). How would this transaction affect Fossils accounting equation? a. Increase both liabilities and stockholders equity...
-
Rescue Sequences LLC purchased inventory' by issuing a $30,000, 10%, 60-day note on October 1. Prepare the journal entries for Rescue Sequences to record the purchase and payment assuming it uses a...
-
The ABC company (an assumed company) is considering moving to a public cloud. The executives would like to assess how this migration would affect their ROI. Help the executives by discussing the...
-
The Harriet Hotel in downtown Boston has 100 rooms that rent for $150 per night. It costs the hotel $30 per room in variable costs (cleaning, bathroom items, etc.) each night a room is occupied. For...
-
Engberg Company installs lawn sod in home yards. The company's most recent monthly contribution format income statement follows: Percent of Sales Variable expenses Contribution margin Fixed expenses...
-
Identify and describe an example of each of the three elements in your organization. the Elements of Organizational Culture as having three elements 1) artifacts, 2) shared values, and 3) shared...
-
Goes short on 10 European put option contracts with a strike price of $108, a quoted option price of $3, the spot price is $107, what's the profit?
-
So basically, there is this couple that invest $50,000 into a managed fund that has equal exposure to equity, bonds (both australian and international), and A-reits. However, the average 10 year...
-
what is The most difficult and important step in risk management process and one that can lead to a risk manager being terminated if it is not conducted properly? Explain and justify.
-
Discuss some of the ways you can conserve electrical energy. Include in your discussion the possible changes that you can bring about in the use of electricity at home.
-
You are a consultant advising a startup business. The business plans to sell a new product with a selling price of $50 per unit. The variable cost per unit is $30, and the fixed costs are $100,000....
-
What are the main distinctions between the different schools of legal interpretation?
-
(a) For the same data and null hypothesis, is the P-value of a one-tailed test (right or left) larger or smaller than that of a two-tailed test? Explain. (b) For the same data, null hypothesis, and...
-
The director of library services at Fairmont College did a survey of types of books (by subject) in the circulation library. Then she used library records to take a random sample of 888 books checked...
-
Let z be a random variable with a standard normal distribution. Find the indicated probability and shade the corresponding area under the standard normal curve. P(z 0.13)
-
Fill in the blank X ~ N(1, 2) = _______
-
The trial balance columns of the worksheet for Briscoe Company at June 30, 2008, are as follows. Other data: 1. A physical count reveals \($300\) of supplies on hand. 2. \($100\) of the unearned...
-
Emil Skoda Company had the following adjusted trial balance. Instructions (a) Prepare closing entries at June 30, 2008. (b) Prepare a post-closing trial balance. EMIL SKODA COMPANY Adjusted Trial...

Study smarter with the SolutionInn App


