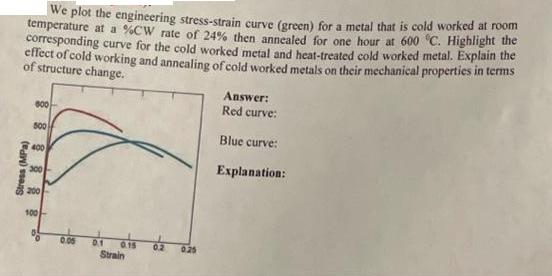
Question: We plot the engineering stress-strain curve (green) for a metal that is cold worked at room temperature at a %CW rate of 24% then
We plot the engineering stress-strain curve (green) for a metal that is cold worked at room temperature at a %CW rate of 24% then annealed for one hour at 600 C. Highlight the corresponding curve for the cold worked metal and heat-treated cold worked metal. Explain the effect of cold working and annealing of cold worked metals on their mechanical properties in terms of structure change. Stress (MPa) 600 500 400 300 200 100 0.05 0.1 0.15 0.2 Strain 0.25 Answer: Red curve: Blue curve: Explanation:
Step by Step Solution
3.39 Rating (165 Votes )
There are 3 Steps involved in it
solution has to produce in two temperature condition 1 At Cold Work Temper... View full answer

Get step-by-step solutions from verified subject matter experts