We've dealt with the molecular diagram of diatomic molecules. However, conceptually the problem is much more...
Fantastic news! We've Found the answer you've been seeking!
Question:
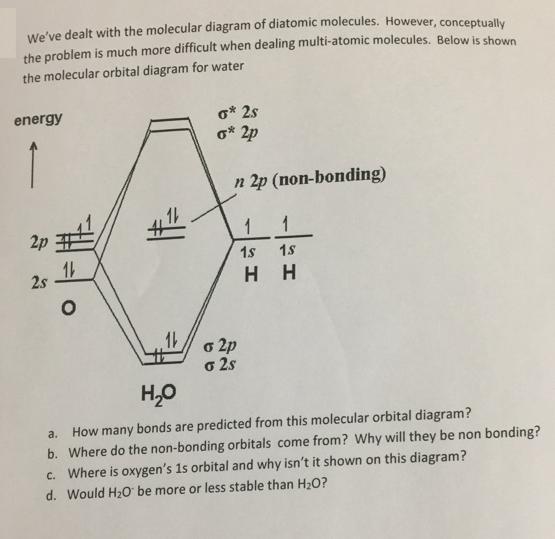
Transcribed Image Text:
We've dealt with the molecular diagram of diatomic molecules. However, conceptually the problem is much more difficult when dealing multi-atomic molecules. Below is shown the molecular orbital diagram for water o* 2s o* 2p energy n 2p (non-bonding) 1 1 2p 1s 18 2s H H o 2p o 2s H,0 How many bonds are predicted from this molecular orbital diagram? b. Where do the non-bonding orbitals come from? Why will they be non bonding? c. Where is oxygen's 1s orbital and why isn't it shown on this diagram? a. d. Would H2o be more or less stable than H2O? We've dealt with the molecular diagram of diatomic molecules. However, conceptually the problem is much more difficult when dealing multi-atomic molecules. Below is shown the molecular orbital diagram for water o* 2s o* 2p energy n 2p (non-bonding) 1 1 2p 1s 18 2s H H o 2p o 2s H,0 How many bonds are predicted from this molecular orbital diagram? b. Where do the non-bonding orbitals come from? Why will they be non bonding? c. Where is oxygen's 1s orbital and why isn't it shown on this diagram? a. d. Would H2o be more or less stable than H2O?
Expert Answer:
Answer rating: 100% (QA)
a four bonds are predicted from this molecular orbital diag... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
How do 2p orbitals differ from each other? How do 2p and 3p orbitals differ from each other? What is a nodal surface in an atomic orbital?
-
When an electron occupies a 2s orbital on an N atom it has a hyperfine interaction of 55.2 mT with the nucleus. The spectrum ofN02 shows an isotropic hyperfine interaction of 5.7 mT. For what...
-
Why do we hybridize atomic orbitals to explain the bonding in covalent compounds? What type of bonds form from hybrid orbitals, or ? Explain.
-
Day and Night formed an accounting partnership in 2014. Capital transactions for Day and Night during 2014 are as follows: Partnership net income for the year ended December 31, 2014; is $68,400...
-
A 2012 survey conducted by Idea Works provided data showing the percentage of seats available when customers try to redeem points or miles for free travel. For each airline listed, the column labeled...
-
(a) At what relative velocity is = 1.50? (b) At what relative velocity is = 100?
-
A double acting reciprocating pump delivering 80 litres per second has the following specifications : Diameter of piston = 300 mm; Stroke = 600 mm; Speed = 80 rpm; Suction head = 5 m; Delivery head =...
-
Following is a trial balance for the Jayce County General Hospital, a governmental hospital. The hospital does not use fund accounting. During 2012, the following transactions took place: 1. Services...
-
1. For the object shown in figure 1,(a) Find the moment of inertia about the x axis.(b) Find the moment of inertia about the y axis.(c) Find the product of inertia.(d) Find the polar moment of...
-
In this mini-case, you will complete the test of details on accounts receivable for the 2019 audit of EarthWear Clothiers, Inc. The principal test of detail involves sending "confirmations" or...
-
If rent per square foot of a property is $15 per year, total rentable square footage is 5,000 square feet and capital expenditure is expected to be $20,000, what is the potential gross income the...
-
If an equation obeys Laplace's equation, then at what angle must lines of constant temperature cross lines of constant flux?
-
Indirect business taxes and capital consumption allowance are not income, yet they are included in the calculation of GDP as income received. Why do we add these two nonincome components to the other...
-
Consider the welding situation of the previous problem. This time let's consider the case where we have convective heat transfer from the rod's surface. For a rod of radius, \(r_{o}\), and a heat...
-
Why dont we measure national output by simply counting the total number of goods and services produced each year?
-
As a biochemical engineer you are evaluating a drug delivery system for an artificial protein to combat Alzheimer's. The protein is very large (200,000 molecular weight) and bulky and is sensitive to...
-
Develop your analysis using the variables in all 5 of the areas : Purpose, Non-verbal, Strategies for success, Style, and Oral strategies and tactics....
-
If a force of F = 50 Ib is applied to the pads at A and C, determine the smallest dimension d required for equilibrium if the spring has an unstretched length of 1 ft. B 1 ft 1 ft F k = 15016/fr 1ft...
-
In an experiment to study the photoelectric effect, a scientist measures the kinetic energy of ejected electrons as a function of the frequency of radiation hitting a metal surface. She obtains the...
-
Write a balanced equation for the reaction of each of the following compounds with water: (a) SO2(g) (b) Cl2O7(g) (c) Na2O2(s) (d) BaC2(s) (e) RbO2(s) (f) Mg3N2(s) (g) NaH(s).
-
Indicate whether each statement is true or false. If it is false, rewrite it so that it is true. (a) If you measure the rate constant for a reaction at different temperatures, you can calculate the...
-
How the duration of a sprint can be decided?
-
Variants and exception handlers are alternate flows for a use case. In which situations should one or the other be used?
-
Why estimating software development effort is so difficult? What are the obstacles?

Study smarter with the SolutionInn App


