What additions must you make to the cell for it to generate astandard EMF? (Check all that
Question:
What additions must you make to the cell for it to generate astandard EMF?
(Check all that apply)
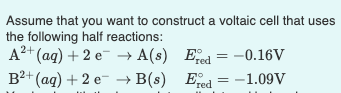
1.) You should apply the initial EMF to the electrode
2.) You should add enough salt containingB+ into the glass containing the electrode B(s) toproduce a 1 M B2+ solution
3.) You should add enough salt containingA2+ into the glass containing the electrode B(s) toproduce a 1 M A2+ solution
4.) You should add enough salt containingB2+ containing the electrode A(s) to produce a 1 MB2+ solution
5.) You should add the salt bridge
6.) You should add enough salt containingA2+ into the glass containing the electrode A(s) toproduce 1 M A2+ solution
Part B:
Which electrode functions as the cathode, and in which directiondo electrons move through the external circuit?
A.) Electrode A functions as the cathode, and electrons flowfrom electrode B to electrode A
B.) Electrode B functions as the cathode, and electrons flowfrom electrode A to electrode B
C.) Electrode A functions as the cathode, and electrons flowfrom electrode A to electrode B
D.) Electrode B functions as the cathode, and electrons flowfrom electrode B to electrode A

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward




