27) Provide the structure of the major organic product in the reaction below. CH 28) Provide...
Fantastic news! We've Found the answer you've been seeking!
Question:
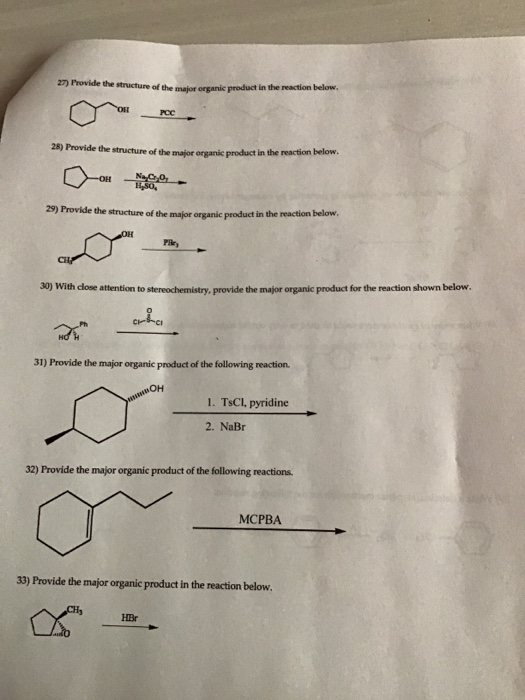
Transcribed Image Text:
27) Provide the structure of the major organic product in the reaction below. CH 28) Provide the structure of the major organic product in the reaction below. Na CO₂ H₂SO, 29) Provide the structure of the major organic product in the reaction below. OH -OH PCC 30) With close attention to stereochemistry, provide the major organic product for the reaction shown below. cha CH₂ Pile, 31) Provide the major organic product of the following reaction. он 32) Provide the major organic product of the following reactions. HBr 1. TsCl, pyridine 2. NaBr 33) Provide the major organic product in the reaction below. MCPBA 27) Provide the structure of the major organic product in the reaction below. CH 28) Provide the structure of the major organic product in the reaction below. Na CO₂ H₂SO, 29) Provide the structure of the major organic product in the reaction below. OH -OH PCC 30) With close attention to stereochemistry, provide the major organic product for the reaction shown below. cha CH₂ Pile, 31) Provide the major organic product of the following reaction. он 32) Provide the major organic product of the following reactions. HBr 1. TsCl, pyridine 2. NaBr 33) Provide the major organic product in the reaction below. MCPBA
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Write the structure of the major organic product obtained by hydroboration-oxidation of each of the following alkenes: (a) 2-Methylpropene (b) cis-2-Butene (c) (d) Cyclopentene (e) 3-Ethyl-2-pentene...
-
A. Draw the structure of the major organic product of the reaction below. B. Either the reactant (X) or the major organic product is missing from the equation below. Draw the missing compound. C....
-
Write the structure of the major organic product isolated from the reaction of 1-hexyne with (a) Hydrogen (2 mol), platinum (b) Hydrogen (1 mol), Lindlar palladium (c) Lithium in liquid ammonia (d)...
-
Write a method to take an integer array as a parameter and return how many elements having the same digits in each element in the array for example if we have array 1 1 , 4 4 , 1 4 , 2 3 , 1 2 , 5 6...
-
Rimes Restaurant makes large cash deposits daily. The bookkeeper recorded one of the daily deposits as $5,700 in the companys records, but the bank statement showed that deposit as $7,500. After...
-
1. Suppose that Sun Farms supplies the basil that survived the storm but the basil does not pass the chemical-residue inspection. Which concept discussed in the chapter might allow Vals to refuse to...
-
Consider the following cash flow profile, and assume MARR is 10 percent/year. a. What does Descartes' rule of signs tell us about the IRR(s) of this project? b. What does Norstrom's criterion tell us...
-
The comparative statements of Lucille Company are presented here. All sales were on account. Net cash provided by operating activities for 2012 was $220,000. Capital expenditures were $136,000, and...
-
You have made a physical pendulum by swinging a rod of mass M = 0.73 kg and length L = 0.53 meters around its end. The mass of the rod is distributed uniformly along its length. We will assume that...
-
A beam of T section is supported and loaded as shown. a) (15 pts) Construct the shear and bending moment diagram, provide expressions for the shear and bending moment on each segment, provide graphs,...
-
Lead thiocyanate, Pb(SCN)2, has a Ksp value of 2.00 x 10-5
-
To ensure messages are not modified, which type of security service would you use? Data Confidentiality Data Integrity Authentication Nonrepudiation
-
The equilibrium constant K for the synthesis of ammonia is 6.8x105 at 298 K. What will K be for the reaction at 375 K? N(g) +3H(g) = 2NH3(g) AH = -92.22 kJ/mol
-
What is 4 days to 9 weeks in ratio and rounding decimal place And what is 8 minutes to 2hours in ratio
-
As shown below, a force of 490 N pushes on a 19 kg box. Starting from rest, the box achieves a velocity of 2.1 m/s in a time of 4.7 s. Find the coefficient of kinetic friction between box and floor....
-
Use the following financial statements to answer Questions 1-16. Sales Cost of goods sold Selling & administrative expenses Depreciation Kramerica Industries (KI) 2016 Income Statement Earnings...
-
Suppose G and H are two groups and p is a prime. (a) Show that if R is a Sylow p-subgroup of G x H, then there is a Sylow p-subgroup P of G and a Sylow p-subgroup Q of H so that R = P x Q. (b) Show...
-
You deposit $10,000 in a savings account that earns 7.5% simple interest per year. What is the minimum number of years you must wait to double your balance? Suppose instead that you deposit the...
-
Predict the product of the reaction of propanal with each of the following: (a) Lithium aluminum hydride (b) Sodium borohydride (c) Hydrogen (nickel catalyst) (d) Methylmagnesium iodide, followed by...
-
Write a structural formula for the CH3NO isomer characterized by the structural unit indicated. None of the atoms in the final structure should have a formal charge. (a) C--N==O (b) C==N--O (c)...
-
The p-toluenesulfonate derived from (R)-2-octanol and ptoluenesulfonyl chloride was allowed to react with sodium benzenethiolate (C6H5SNa). Give the structure, including stereochemistry and the...
-
Based on Exhibit 1, the fi nancial leverage ratio for Company B is closest to: A . 0.55. B . 1.22. C . 2.22. EXHIBIT 1 Common-Size Balance Sheets for Company A, Company B, and Sector Average Company...
-
Based on Exhibit 1, which ratio indicates lower liquidity risk for Company A compared with Company B? A . Cash ratio. B . Quick ratio. C . Current ratio. EXHIBIT 1 Common-Size Balance Sheets for...
-
Under which section of a manufacturing companys cash fl ow statement are the following activities reported? Item 1: Purchases of securities held for trading Item 2: Sales of securities considered...

Study smarter with the SolutionInn App



