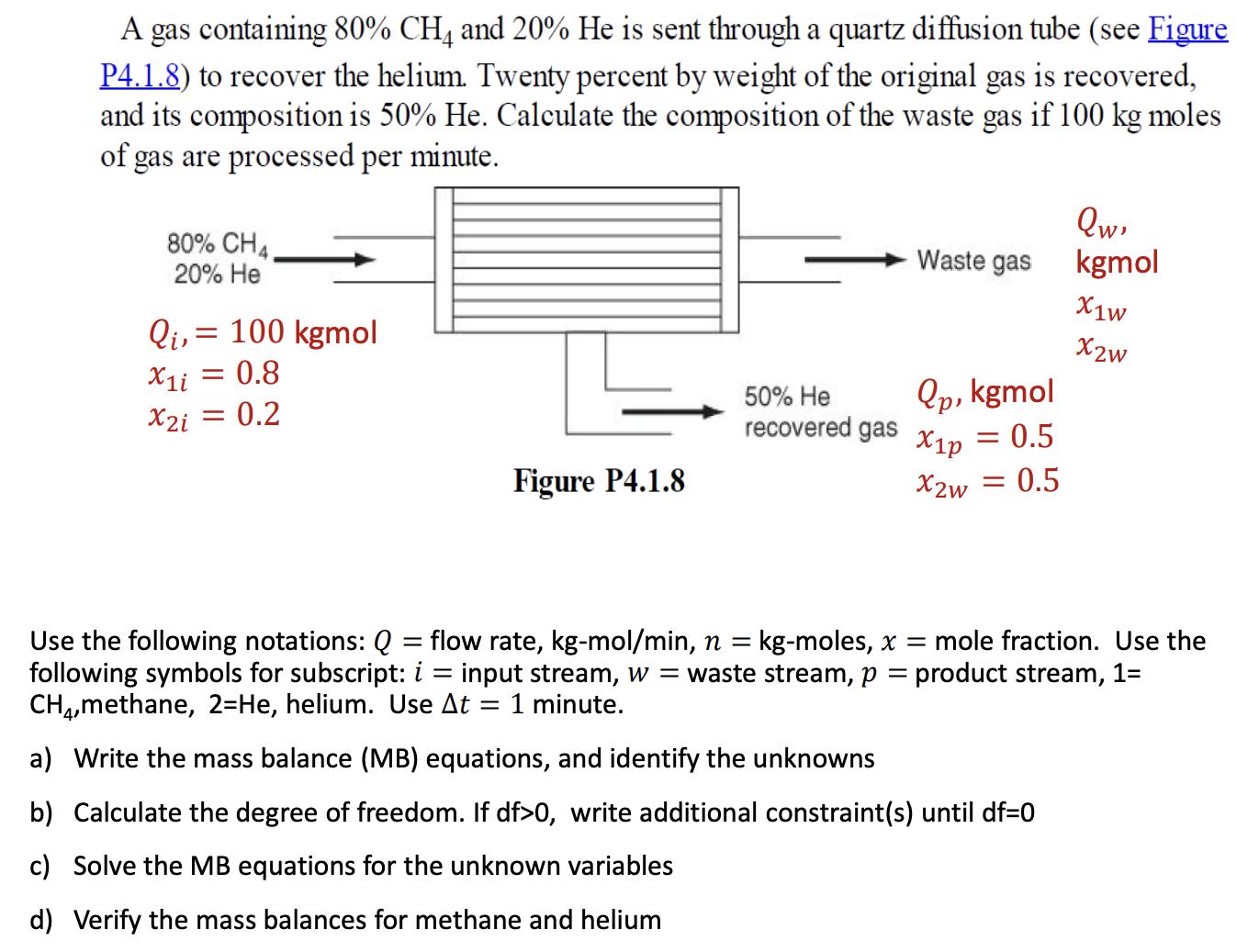
A gas containing 80% CH4 and 20% He is sent through a quartz diffusion tube (see...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A gas containing 80% CH4 and 20% He is sent through a quartz diffusion tube (see Figure P4.1.8) to recover the helium. Twenty percent by weight of the original gas is recovered, and its composition is 50% He. Calculate the composition of the waste gas if 100 kg moles of gas are processed per minute. 80% CH4, 20% He Qi, = 100 kgmol X1i = 0.8 X2i = 0.2 Figure P4.1.8 - 50% He recovered gas Waste gas Qp, kgmol = 0.5 X1p X2w = 0.5 Qw, kgmol X1w X2w Use the following notations: Q = flow rate, kg-mol/min, n = kg-moles, x = mole fraction. Use the following symbols for subscript: i = input stream, w = waste stream, p = product stream, 1= CH4,methane, 2-He, helium. Use At 1 minute. a) Write the mass balance (MB) equations, and identify the unknowns b) Calculate the degree of freedom. If df>0, write additional constraint(s) until df=0 c) Solve the MB equations for the unknown variables d) Verify the mass balances for methane and helium A gas containing 80% CH4 and 20% He is sent through a quartz diffusion tube (see Figure P4.1.8) to recover the helium. Twenty percent by weight of the original gas is recovered, and its composition is 50% He. Calculate the composition of the waste gas if 100 kg moles of gas are processed per minute. 80% CH4, 20% He Qi, = 100 kgmol X1i = 0.8 X2i = 0.2 Figure P4.1.8 - 50% He recovered gas Waste gas Qp, kgmol = 0.5 X1p X2w = 0.5 Qw, kgmol X1w X2w Use the following notations: Q = flow rate, kg-mol/min, n = kg-moles, x = mole fraction. Use the following symbols for subscript: i = input stream, w = waste stream, p = product stream, 1= CH4,methane, 2-He, helium. Use At 1 minute. a) Write the mass balance (MB) equations, and identify the unknowns b) Calculate the degree of freedom. If df>0, write additional constraint(s) until df=0 c) Solve the MB equations for the unknown variables d) Verify the mass balances for methane and helium
Expert Answer:
Answer rating: 100% (QA)
A racemic mixture refers to a mixture which consists of equal amounts of two enantiomers This means ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
What is a racemic mixture? Describe one method of resolving a racemic mixture.
-
The sugar concentration in a solution (e.g., in a urine specimen) can be measured conveniently by using the optical activity of sugar and other asymmetric molecules. In general, an optically active...
-
Googles ease of use and superior search results have propelled the search engine to its num- ber one status, ousting the early dominance of competitors such as WebCrawler and Infos- eek. Even later...
-
Identify each of the equations as representing either a circle, a parabola, an ellipse, a hyperbola, or none of these. (x + 1) 2 + (y + 1) 2 = 2(x + y + 1)
-
King Company leased equipment from Mann Industries. The lease agreement qualifies as a finance lease and requires annual lease payments of $52,538 over a six-year lease term (also the asset's useful...
-
The titration of Na2CO3 with HCl has the following qualitative profile: pH Vi mL HCI
-
Bats are capable of navigating using the earth's field-a plus for an animal that may fly great distances from its roost at night. If, while sleeping during the day, bats are exposed to a field of a...
-
Lang Drug needs to determine the proper capacity level for a new drug, Niagara. Its goal is to maximize the expected NPV earned from the drug during years 0-14, assuming a discount rate of 10 percent...
-
Describe Protection of depositor's funds in the US, UK and Bahamas.
-
Big Tree Lumber has earnings per share of $1.36. The firm's earnings have been increasing at an average rate of 2.9 percent annually and are expected to continue doing so. The firm has 21,500 shares...
-
Nova Inc. has liabilities of 10 due in 1, 4, and 7 years. What asset incomes must the company arrange for in years 1 and 6 to immunize their cash flow, assuming an annual interest rate of 10% on all...
-
An investor considers whether to invest in debt or equity of LIPPBORG PLC. Since he already has to pay a high personal tax rate, he does not want to pay more taxes than necessary. Therefore, he...
-
Build Your Budget Instructions: You are going to think about everything that your program needs, or wishes to have. You are going to use the word document to plan out your yearly budget, estimating...
-
Suppose yields at the long end of the Treasury curve: Issue Yield to Maturity 1.625% due Aug 2031 1.711% 2.88% 3% due Aug 2051 Modified Duration (in yrs) 9.077 19.474 a. How would you, in 2021, bet...
-
You are employed as a network consultant at Computer Systems and Networks Design Consultant. An e-commerce business organisation is going through a major transformation because of its expansion. The...
-
The production of the Reliable Manufacturing Company for 2013 and part of 2014 is as follows: 2013 Production (thousands) 2014 Production (thousands) 2013 Production (thousands) 2014 Production...
-
Consider the discrete group G of order 8 that has the following Cayley diagram e If we have the sequence of operations: fcagec, which of the options represents the reduction of the sequence to a...
-
Describe the formation of a covalent bond in H2 from atoms. What does it mean to say that the bonding electrons are shared by the two atoms?
-
A 0.150 M solution of NaClO is prepared by dissolving NaClO in water. A 50.0-mL sample of this solution is titrated with 0.100 M HCl. Calculate the pH of the solution at each of the following points...
-
What is the most important commercial means of producing oxygen?
-
Consider a situation in which you do not know the timing or amounts of individual cash flows. However, you do know the discounted and summed values of the revenues, \(\sum R t^{*}(P \mid F, M A R R,...
-
True or False: Benefits and disbenefits must be converted to monetary values to use benefit-cost analysis.
-
Elm City is considering a replacement for its police radio. The benefits and costs of the replacement are shown below. What is the replacement's benefit/cost ratio if the effective annual interest...

Study smarter with the SolutionInn App


