What is the correct formula for the precipitate produced after aqueous solutions of magnesium nitrate and...
Fantastic news! We've Found the answer you've been seeking!
Question:
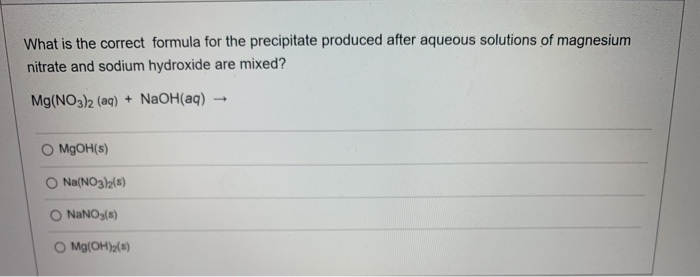
Transcribed Image Text:
What is the correct formula for the precipitate produced after aqueous solutions of magnesium nitrate and sodium hydroxide are mixed? Mg(NO3)2 (aq) + NaOH(aq) O MgOH(s) O Na(NO3)2(s) NaNO3(s) Mg(OH)₂(s) - What is the correct formula for the precipitate produced after aqueous solutions of magnesium nitrate and sodium hydroxide are mixed? Mg(NO3)2 (aq) + NaOH(aq) O MgOH(s) O Na(NO3)2(s) NaNO3(s) Mg(OH)₂(s) -
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the correct way to annualize an interest rate in financial decision making?
-
Which one of the following is the correct formula for EVA? a. Net operating profit before tax (Cost of capital Invested capital) b. Net operating profit after tax (Cost of capital Invested...
-
Consider a 1.50- g mixture of magnesium nitrate and magnesium chloride. After dissolving this mixture in water, 0.500 M silver nitrate is added dropwise until precipitate formation is complete. This...
-
Consider an ideal dual-loop heat-powered refrigeration cycle using R-134a as the working fluid, as shown in Fig. P9.135. Saturated vapor at 200 F leaves the boiler and expands in the turbine to the...
-
Consider three securities: a. A floating-rate bond. b. A preferred share paying a fixed dividend. c. A floating-rate preferred. If you were responsible for short-term investment of your firms excess...
-
An outlier can ____ the value of a correlation.
-
Environmental Concerns Limited (ECL) issued \$500,000 of 10-year, 6\% bonds payable at maturity (par) value on May 1, 2008. The bonds pay interest each April 30 and October 31, and the company ends...
-
State whether or not a trust is created in each of the following situations: (a) A declares herself trustee of the bulk of my securities in trust for B. (b) A, the owner of Blackacre, purports to...
-
Why governments should not implement wealth redistribution measures and limit corporate wealth to address inequality and curb the significant poverty that exists not only in Canada but across the...
-
This is the code followed by the questions :) // Execute in strict mode to prevent some common mistakes "use strict"; /** * Make diary data item * @param type Type of item to create, either "text" or...
-
A plant has three linked genes, ABC, however the gene order is unknown. The following cross is performed. Abc/Abc X aBC/aBC. The F1 individuals are crossed to a tester plant. You count 100 progeny...
-
Brief overview of the two models: Traditional Insurance-Based Practice and Direct Primary Care Model.
-
How do advancements in technology and digital media impact the creation, dissemination, and preservation of ethnic art forms, particularly in indigenous and diasporic communities ? Explain
-
replaces characters at indices indx to indx+num-1 with a copy of subStr #include #include using namespace std; int main() { string passwdStr; int idx; cin >> passwdStr; idx = 0; /* Your code goes...
-
Under US GAAP, how is Construction in Progress (CIP) typically classified and reported for long-term contracts?
-
In a salary salary range with minimum of $70,000 with a midpoint of $60,000, an employee with a current salary of $53,000 have a compa-ratio of __________. Use this formula to calculate it: Equation:...
-
People at the top of an organization often delegate the work. Those at the bottom are usually working based on the clock - work 8 - 5 with lunch at noon and two breaks. It is the middle management...
-
Software Solution is family-owned business that has been in operation for more than 15 year. The board of directors is comprised of mainly family members, plus a few professionals such as an...
-
Calculate the percentage composition for each of the following compounds (three significant figures). a. NO b. SO 3 c. NaH 2 PO 3 d. Pb(NO 3 ) 2
-
The energy of dissociation of the Cl 2 molecule is 240 kJ/mol. Using the appropriate data from this chapter, calculate the energy obtained (or released) in adding electrons to the Cl 2 molecule to...
-
A sample of an ionic solid is dissolved in 1.00 kg of water. The freezing point of the water is 0.01C. If three times the mass of ionic solid is dissolved in 1.00 kg of water and the resulting...
-
A W14 \(\times 30\) structural A992 steel column is pin connected at is ends and has a length \(L=12 \mathrm{ft}\). Determine the maximum eccentric load \(P\) that can be applied so the column does...
-
A W16 \(\times 45\) structural A992 steel column is fixed at the base and free at the top and has a length \(L=8 \mathrm{ft}\). Determine the maximum eccentric load \(P\) that can be applied so the...
-
Determine the \(\operatorname{load} P\) required to cause the steel \(\mathrm{W} 12 \times 50\) structural A-36 steel column to fail either by buckling or by yielding. The column is fixed at its...

Study smarter with the SolutionInn App



