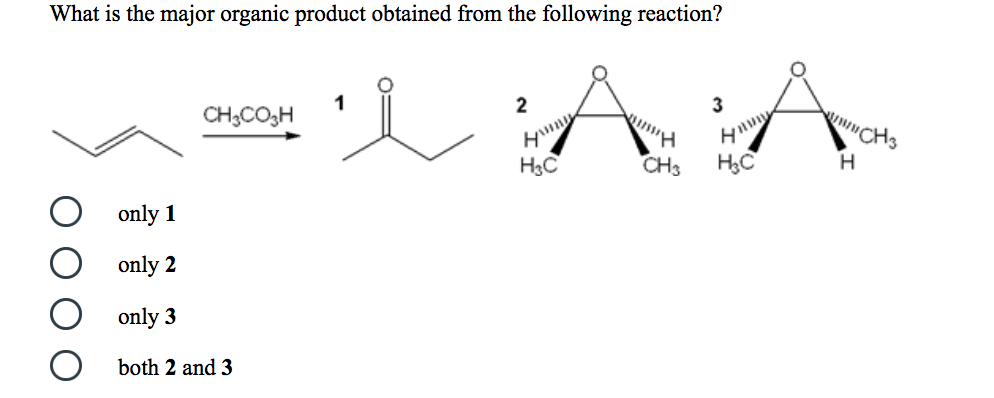
What is the major organic product obtained from the following reaction? CHCO3H only 1 only 2...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
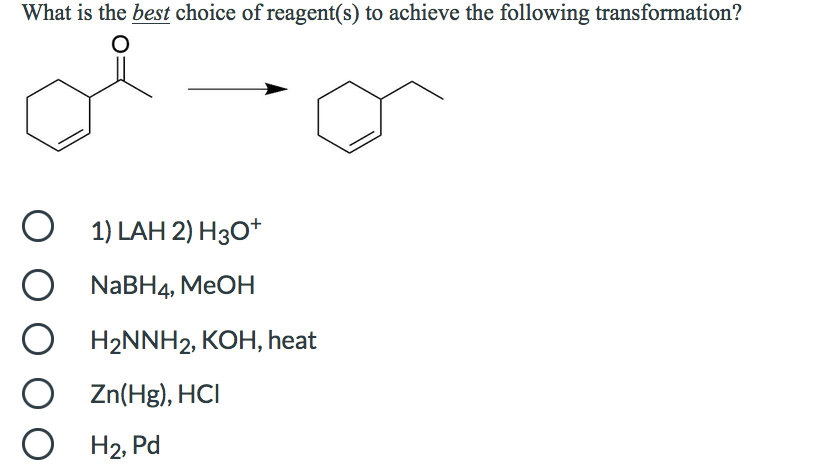
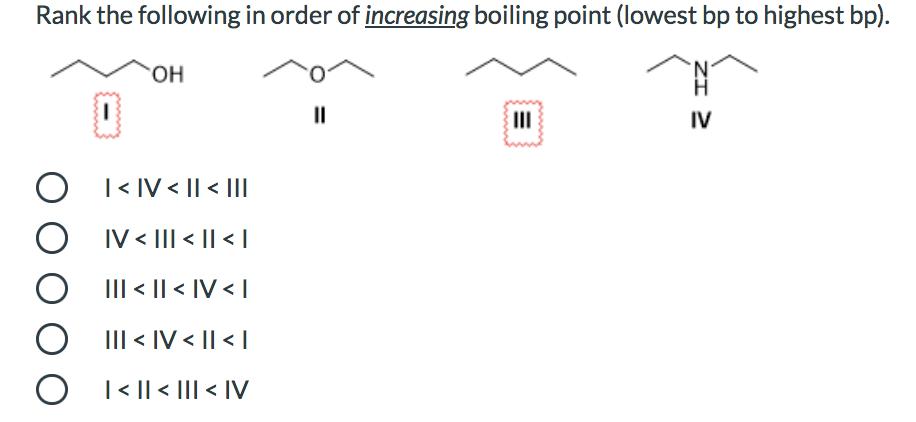
What is the major organic product obtained from the following reaction? CH₂CO3H only 1 only 2 only 3 both 2 and 3 1 A A Hinny H₂C CH3 H₂C CH 3 What is the best choice of reagent(s) to achieve the following transformation? O 1) LAH 2) H30+ O NaBH4, MeOH O H₂NNH2, KOH, heat Zn(Hg), HCI H₂, Pd O Rank the following in order of increasing boiling point (lowest bp to highest bp). O O O O OH |< IV < || < ||| IV < ||| < || < | ||| < || < IV < | III < IV < || < | | < || < ||| < |V || III IV What is the major organic product obtained from the following reaction? CH₂CO3H only 1 only 2 only 3 both 2 and 3 1 A A Hinny H₂C CH3 H₂C CH 3 What is the best choice of reagent(s) to achieve the following transformation? O 1) LAH 2) H30+ O NaBH4, MeOH O H₂NNH2, KOH, heat Zn(Hg), HCI H₂, Pd O Rank the following in order of increasing boiling point (lowest bp to highest bp). O O O O OH |< IV < || < ||| IV < ||| < || < | ||| < || < IV < | III < IV < || < | | < || < ||| < |V || III IV
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below Your question appears fragmented There are three separate images with different questions ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
What is the major organic product obtained from the following reaction? NaOCH, CH3OH
-
What is the major organic product obtained from the following reaction? 1. KCN 2. H*
-
Write the structure of the major organic product obtained by hydroboration-oxidation of each of the following alkenes: (a) 2-Methylpropene (b) cis-2-Butene (c) (d) Cyclopentene (e) 3-Ethyl-2-pentene...
-
The simple truss shown has the following dimensions: H1 = 1.90 in; H2 = 1.60 in; L0=31 in; L2 = 14 in; D1 = 0.76 in; D2 = 0.86 in. Both supporting bars have a depth of 0.5 in. If the maximum...
-
For each of the following accounts, determine its classification on the balance sheet using the following classifications. Use OOE for other owners equity items that are not contributed capital or...
-
Based on the data presented in Exercise 6-23, journalize Boyle Co.s entries for (a) the purchase, (b) the return of the merchandise for credit, and (c) the payment of the invoice within the discount...
-
The velocity potential for a certain inviscid, incompressible flow field is given by the equation \[ \phi=2 x^{2} y-\left(\frac{2}{3} ight) y^{3} \] where \(\phi\) has the units of \(\mathrm{m}^{2} /...
-
The lead time for KraftyCity workbenches is 3 weeks, with a standard deviation of 1.2 weeks, and the average weekly demand is 24, with a standard deviation of 8 work benches. What should the reorder...
-
Geneva Conventions are customary approved agreements for international treaties that was created in Geneva Switzerland between 1864 and 1949. These treaties serve as a guide to restrain unsparing...
-
Roman Systems Inc. (RSI) is a Canadian private company. It was incorporated in Year 1 by its sole common shareholder, Marge Roman. RSI manufactures, installs, and provides product support for its...
-
Write a C Program for recognizing identifiers and delimiters from a large string.
-
Are perfectly competitive firms price makers or price takers? How is the profit-maximizing price and quantity decided upon by the perfectly competitive firm?
-
If a Fixed Based Operator (FBO) charges $5 per gallon for jet fuel with a marginal cost of $4, what is the markup on cost? When Ep = 2, the optimal markup on cost is:
-
Many airlines overbook flights. Is this efficient from an airlines point of view?
-
When the price of airline tickets falls, the supply falls and the demand increases. Using the economic ideas covered in this chapter, critically evaluate this statement.
-
How were airline ticket prices set during the period of regulation? From an economic point of view, what would be the predicted outcome of setting prices in this way?
-
Question 2 Assume that a firm involves on a competitive market. There are 50 firms. Total Cost 130 175 Quantity 01234567 7 8 9 205 235 255 280 310 350 410 490 Variable Cost 130 Marginal Cost 15000LC...
-
For liquid water the isothermal compressibility is given by; where r and b are functions of temperature only. If 1 kg of water is compressed isothermally and reversibly from I to 500 bar at 60(C. how...
-
Refer to the molecular orbital diagrams of allyl cation (Figure 10.12) and those presented earlier in this chapter for ethylene and 1,3-butadiene (Figures 10.8 and 10.9) to decide which of the...
-
Give a structural formula for the carbocation intermediate that leads to the major product in each of the reactions of Problem 6.3 (Section 6.5). In Problem 6.3 Write the structure of the major...
-
An article in the October 1998 issue of the Journal of Chemical Education (p. 1266) describes the following reaction. Fluoxetine hydrochloride (Prozac) is a widely prescribed antidepressant drug...
-
Determine the static deflections in each of the springs in the system of Figure P2.22. 40 cm 20 cm 1 x 105 N/m 2 x 105 N/m FIGURE P 2.22 4 kg
-
A \(30 \mathrm{~kg}\) compressor sits on four springs, each of stiffness \(1 \times 10^{4} \mathrm{~N} / \mathrm{m}\). What is the static deflection of each spring.
-
The propeller of a ship is a tapered circular cylinder, as shown in Figure P2.24. When installed in the ship, one end of the propeller is constrained from longitudinal motion relative to the ship...

Study smarter with the SolutionInn App


