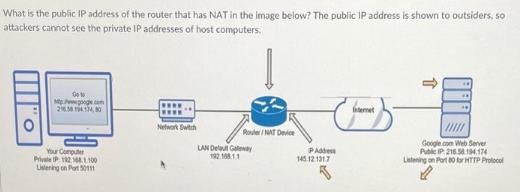
What is the public IP address of the router that has NAT in the image below?...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
What is the public IP address of the router that has NAT in the image below? The public IP address is shown to outsiders, so attackers cannot see the private IP addresses of host computers. O ON Mtp://www.google.com 216.584124,80 Your Computer Private IP 192.168.1.100 Listening on Port50111 wwww Network Switch Router/NAT Device LAN Delaut Galway PAdde 145.12.131.7 met Google.com Web Server Public IP: 216.58.194.174 Listening on Part 80 for HTTP Pro What is the public IP address of the router that has NAT in the image below? The public IP address is shown to outsiders, so attackers cannot see the private IP addresses of host computers. O ON Mtp://www.google.com 216.584124,80 Your Computer Private IP 192.168.1.100 Listening on Port50111 wwww Network Switch Router/NAT Device LAN Delaut Galway PAdde 145.12.131.7 met Google.com Web Server Public IP: 216.58.194.174 Listening on Part 80 for HTTP Pro
Expert Answer:
Answer rating: 100% (QA)
The public IP address of the router is 145121317 This is the ... View the full answer

Related Book For 

Business Data Networks and Security
ISBN: 978-0134817125
11th Edition
Authors: Raymond R. Panko, Julia L. Panko
Posted Date:
Students also viewed these computer network questions
-
How do psychosocial factors such as job demands, control, support, and reward influence the experience and perception of stress in the workplace, and what strategies can organizations employ to...
-
A network has to be designed for a small business organization which has 100 users. The organization hosts an e-commerce application on a server which is accessible to internet users using https and...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Explain why entrepreneurial firms are often in a strong position to use combination strategies.
-
What is the chemical composition of petroleum?
-
Wharton, Inc. pays income taxes on capital gains at a rate of 30 percent. At December 31, year 1, the company owns marketable securities that cost $180,000 but have a current market value of...
-
Figure 11.65 presents the PFD of a process for the recovery of methane from a feed stream consisting of \(75 \%\) methane and \(25 \%\) nitrogen. The process feed is first cooled from 90 to...
-
Jackie Wilson, manager of Expert Building Company, is a valued and trusted employee. She has been with the company from its start two years ago. Because of the demands of her job, she has not taken a...
-
Presented below is information related to the purchases of common stock by Nash Company during 2025. Cost Fair Value (at December 31) (at purchase date) Investment in Arroyo Company stock $90,000...
-
The points A(0, 0), B(0.5, 0.75), C(0.8, 1.44), D(0.95, 1.8525), E(0.99, 1.9701) and F(1, 2) lie on the curve y = f(x). a. Copy and complete the table to show the gradients of the chords CF, DF and...
-
Find the standard deviation for the following set of data: 98, 75, 85, 91 Question 2 Find the standard deviation for the following set of data: 47, 5, 28, 91 Question 3 1 pts 1 pts If you have a set...
-
A company has 1 0 employees who each work 2 , 0 0 0 hours per year. If practical capacity per employee is 8 5 % , the number of minutes per employee used in TDABC calculations is ?
-
Have you ever participated in or witnessed a group presentation in which one or two members did not complete their portion of the work? What was that experience like? What steps may group members...
-
2.) Jack places an amount of $ 60 in an account at the end of each year for 12 consecutive years beginning one year from today. If the interest rate is 3 %, how much will Jack have at the end of the...
-
Carbon dioxide from the air dissolves in water, forming small amounts of carbonic acid, H 2 CO 3 , and causes the water to be slightly acidic. Would this tend to make the titration of NaOH and HCL...
-
A band signs a deal in which it receives $26,000/year in perpetuity. If the average inflation rate into the indefinite future is 4.3%, calculate the real total of this income stream. Round your...
-
Which of the following is an internal control procedure used to safeguard a company's assets? Multiple Choice Depositing cash receipts in a bank on a timely basis O Segregation of duties O Preparing...
-
The following selected accounts and normal balances existed at year-end. Notice that expenses exceed revenue in this period. Make the four journal entries required to close the books: Accounts...
-
a. Complete the traffic table in Figure 3-13. b. In Figure 3-12, add 392 Mbps of traffic for Seattle-Ogden communication. Using a picture like the one in the figure, show your work. c. Do it again...
-
a. Distinguish between application message fragmentation and packet fragmentation. b. Under what circumstances would the identification, flags, and fragment offset fields be used in IP? 15b.) The...
-
a. How is the syntax of Ethernet frames depicted? 20a1.) "Octet" is the same as _______. a. "bit" b. "byte" c. Either A or B, depending on the context d. Neither A nor B 20a2.) Ethernet syntax is...
-
In a vapour compression refrigeration system, the condition of refrigerant before entering the compressor is (a) saturated liquid (b) wet vapour (c) dry saturated liquid (d) superheated vapour
-
During a refrigeration cycle, heat is rejected by the refrigerant in (a) compressor (b) condenser (c) evaporator (d) expansion valve
-
The highest temperature during the cycle in vapour compression refrigeration system occurs after (a) compression (b) condensation (c) expansion (d) evaporation

Study smarter with the SolutionInn App


