What orbitals are used to form each bond in methanol, CH3OH? Be sure to answer all...
Fantastic news! We've Found the answer you've been seeking!
Question:
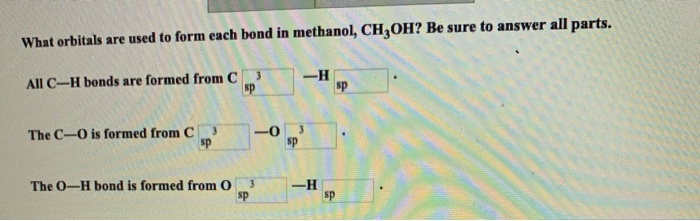
Transcribed Image Text:
What orbitals are used to form each bond in methanol, CH3OH? Be sure to answer all parts. All C-H bonds are formed from C sp The C-O is formed from C 3 sp 3 The O-H bond is formed from O sp -0 3 -H 3 sp -H sp sp What orbitals are used to form each bond in methanol, CH3OH? Be sure to answer all parts. All C-H bonds are formed from C sp The C-O is formed from C 3 sp 3 The O-H bond is formed from O sp -0 3 -H 3 sp -H sp sp
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What words are used to form each of the following acronyms? (a) DBMS (b) DDL (c) DML (d) SQL (e) OLAP (f) OODB
-
What orbitals are used to form the carbon-carbon bond between the highlighted carbons? a. CH3CH==CHCH3 b. CH3CH== CHCH3 c. CH3CH == C == CH2 d. CH3== C== CCH3 e. CH3C==CCH3 f. CH2==CHCH==CH2 g....
-
What orbitals are used to form the 10 covalent bonds in propane (CH 3 CH 2 CH 3 )?
-
Write the expression as one ratio without any negative exponents. x1/4x-3/4 X
-
Gleed Company manufactures products Alpha, Beta, and Gamma from a joint process. Production, sales, and cost data for July follow. Required: 1. Assuming that joint costs are allocated using the...
-
One of your colleagues, Katina Vander, was recently promoted to department manager and now serves on the company's strategic planning committee. At its monthly meeting next week, the committee will...
-
Rank the following four devices by the energy they use for the time period given, from highest to lowest: A. Electric blanket (60 W) used for 8 h B. Flat-screen television (120 W) for 3 h C....
-
The Thomas Company is in the process of developing a revolutionary new product. A new division of the company was formed to develop, manufacture, and market this product. As of year- end (December...
-
What are the key factors when designing products or services for optimal supply chain efficiency? How does product design affect supply chain sustainability? Process Technology: How can advances in...
-
Can you make the journal entries for the transactions below: Dec. 1 - Sold merchandise to Argem Day Care Center for P20,000 and received a 3-month, 12% note. 2 - Purchased merchandise from Stephen...
-
A journal entry to represent the cash payment of $50 to a supplier on June 1st might look like this for cash accounting - 6/1 Dr. Supplies $50 Cr. Cash $50 Assume the supplies were delivered on 5/1...
-
Consider the following system. 2x-3y=6 5x-4y=8 CIS When solving this system by the addition method, eliminate y by multiplying the first equation by 4 and the second equation by and then adding the...
-
Six gallons of a salt solution consists of 35 % salt. It is the result of mixing a 40 % solution with a 30 % solution. How many gallons of each of the solutions was used? Let x = the number of...
-
A free-falling object has a speed of 30 m/s at one instant. Exactly 2 s later its speed will be : a) About 40 m/s b) About 50 m/s. c) The same. d) About 60 m/s
-
Jennifer, who has a mass of 50.0 kg, is riding at 35.0 m/s in her red sports car when she must suddenly slam on the brakes to avoid hitting a deer crossing the road. She strikes the air bag, that...
-
Starting from your campsite you walk 3 . 0 km east, 3 . 0 km north, 2 . 0 km east, and then 1 . 0 km west. What is your final distance from the campsite?
-
A pension plan would like to shield its liabilities, due in exactly 3 years, from interest rate fluctuations. The current YTM is 10%. The strategies it is considering are: a. Purchase a 10-year bond...
-
1. Following are information about Alhadaf Co. Cost incurred Inventory Purchases Sales Adverting expense Salary Expense Depreciation Beginning Inventory Ending Inventory Amount 118,000 350.000 90,000...
-
Tell whether each of the following compounds has the E or the Z configuration: a. b. c. d. H3C CH2CH3 C=C CH3CH2 CH2CH2CI
-
Using the given starting material, any necessary inorganic reagents, and any carbon containing compounds with no more than two carbon atoms, indicate how the following syntheses could be carried out:...
-
Which will have a higher percentage of the diequatorial-substituted conformer, compared with the diaxial-substituted conformer: trans-1,4-dimethylcyclohexane or cis-1-tert-butyl- 3-methylcyclohexane?
-
Define bank capital and key measures of bank profits and returns.
-
Identify the types and sources of bank risk and explain how to control them.
-
Consider a bank with the following balance sheet, as shown on the next page. You read online that the banks return on assets (ROA) was 1 percent. What were the banks after-tax profits? Bank Balance...

Study smarter with the SolutionInn App



