Which of the following acids is/ are used in the reaction of ethanol to form ethene...
Fantastic news! We've Found the answer you've been seeking!
Question:
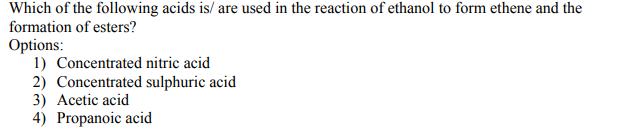
Transcribed Image Text:
Which of the following acids is/ are used in the reaction of ethanol to form ethene and the formation of esters? Options: 1) Concentrated nitric acid 2) Concentrated sulphuric acid 3) Acetic acid 4) Propanoic acid Which of the following acids is/ are used in the reaction of ethanol to form ethene and the formation of esters? Options: 1) Concentrated nitric acid 2) Concentrated sulphuric acid 3) Acetic acid 4) Propanoic acid
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which of the following acids would be most suitable for preparing a buffer of pH 3.10? (i) hydrogen peroxide; (ii) propanoic acid; (iii) cyanoacetic acid; (iv) 4 - aminobenzenesulfonic acid.
-
Which of the following are probability distributions? Why? (a) RANDOM VARIABLE X PROBABILITY 2 ................... 0.1 1 ................. 0.2 0 ................... 0.3 1 ................... 0.25 2...
-
Which of the following are independent demand inventory items? Dependent demand inventory items? a. Bicycles in a toy store b. Bicycle wheels in a bicycle factory c. Blood at a blood bank d....
-
H. S. Black is noted for developing a negative feedback amplifier in 1927. Often overlooked is the fact that three years earlier he had invented a circuit design technique known as feed forward...
-
Find the x- and y-components of vector R, which has a length of 13.0 cm at 30.0.
-
The symptom score of a clinical trial subject given the extended-release form of the prescription drug remained unchanged for 8 hours. Based on Figure 2, the 8-hour period most likely began: A. 3...
-
The dimensions of the rectangular loop of wire in Figure P29.35 are \(\ell=400 \mathrm{~mm}\) and \(w=120 \mathrm{~mm}\). The mass of the loop is \(10 \mathrm{~g}\), its resistance is \(5.0...
-
Now Glinda and Elphaba are even more confused than before. They have three distinct options: to create a package on their own, buy commercial off-the-shelf software such as QuickBooks Pro, or...
-
a) Explain the following terms as used in real estate appraisals i. Value in use ii. iii. Liquidation value Insurable value b) Describe the cost approach of real estate valuation c) Explain the...
-
A nursing home employs attendants who are needed around the clock. Each attendant is paid the same, regardless of when his or her shift begins. Each shift is 8 consecutive hours. Shifts begin at 6...
-
Hudson Corporation will pay a dividend of $5.40 per share next year. The company pledges to increase its dividend by 3.00 percent per year indefinitely. If you require a return of 7.20 percent on...
-
Empire Industries forecasts net income this coming year as shown below (in thousands of dollars): Approximately $150,000 of Empires earnings will be needed to make new, positive- NPV investments....
-
NatNah, a builder of acoustic accessories, has no debt and an equity cost of capital of 17%. NatNah decides to increase its leverage to maintain a market debt-to-value ratio of 0.4. Suppose its debt...
-
Acort Industries owns assets that will have an 70% probability of having a market value of $52 million in one year. There is a 30% chance that the assets will be worth only $22 million. The current...
-
Hawar International is a shipping firm with a current share price of $4.56 and 9.4 million shares outstanding. Suppose that Hawar announces plans to lower its corporate taxes by borrowing $19.7...
-
Marpor Industries has no debt and expects to generate free cash flows of $17 million each year. Marpor believes that if it permanently increases its level of debt to $30.43 million, the risk of...
-
Suppose you have $1850 in your savings account at the end of a certain period of time. You invested $1500 at a 2.34% simple annual interest rate. How long, in years, was your money invested?
-
Express mass density in kg/m3 and weight density in lb/ft3. 1. Find the mass density of a chunk of rock of mass 215 g that displaces a volume of 75.0 cm3 of water. 2. A block of wood is 55.9 in. x...
-
Cu + reacts with neocuproine to form the colored complex (neocuproine)2Cu + , with an absorption maximum at 454 nm. Neocuproine is particularly useful because it reacts with few other metals. The...
-
Detection limit. A sensitive chromatographic method was developed to measure sub-part per-billion levels of the disinfectant by-products iodate (IO 3 ), chlorite (ClO 2 ), and bromate (BrO 3 ) in...
-
Here is an immunoassay to measure explosives such as trinitrotoluene (TNT) in organic solvent extracts of soil. The assay employs a flow cytometer, which counts small particles (such as living cells)...
-
When you were looking at goodwill in the previous chapter, you were interested in the difference between the amount received and the value of net assets. Why do we consider only the assets when there...
-
Fitch and Wall have been in partnership for many years sharing profits and losses in the ratio 5:3 respectively. The following was their statement of financial position as at 31 December 2016: On 1...
-
Alan, Bob and Charles are in partnership sharing profits and losses in the ratio 3:2:1 respectively. The statement of financial position for the partnership as at 30 June 2016 is as follows: Charles...

Study smarter with the SolutionInn App



