Which of the following aqueous solutions has the highest osmotic pressure at 25C (assume 100% dissociation...
Fantastic news! We've Found the answer you've been seeking!
Question:
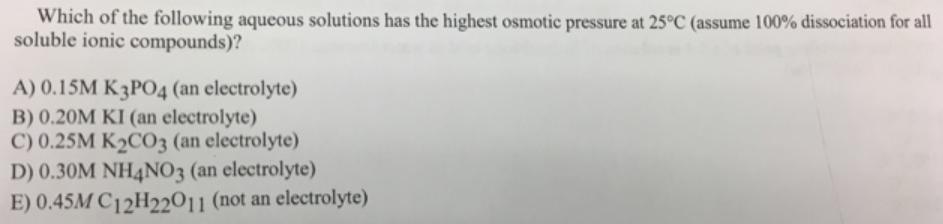
Transcribed Image Text:
Which of the following aqueous solutions has the highest osmotic pressure at 25°C (assume 100% dissociation for all soluble ionic compounds)? A) 0.15M K3PO4 (an electrolyte) B) 0.20M KI (an electrolyte) C) 0.25M K2CO3 (an electrolyte) D) 0.30M NH4NO3 (an electrolyte) E) 0.45M C12H22011 (not an electrolyte) Which of the following aqueous solutions has the highest osmotic pressure at 25°C (assume 100% dissociation for all soluble ionic compounds)? A) 0.15M K3PO4 (an electrolyte) B) 0.20M KI (an electrolyte) C) 0.25M K2CO3 (an electrolyte) D) 0.30M NH4NO3 (an electrolyte) E) 0.45M C12H22011 (not an electrolyte)
Expert Answer:
Answer rating: 100% (QA)
TT icsT T asmotic pressure iz vorit hoff factor Cmolasity S 00821 atm mo T Tem... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Which of the following aqueous solutions has (a) The higher boiling point, (b) The higher freezing point, and (c) The lower vapor pressure: 0.35 m CaCl2 or 0.90 m urea? Explain. Assume CaCl2 to...
-
Which of the following aqueous solutions would you expect to be the best conductor of electricity at 258C? Explain your answer. (a) 0.20 M NaCl (b) 0.60 M CH3COOH (c) 0.25 M HCl (d) 0.20 M Mg(NO3)2
-
Which of the following aqueous solutions has the highest pH and which has the lowest? a. 0.1 M NH3; b. 0.1 M NH4Br; c. 0.1 M NaF; d. 0.1 M NaCl
-
Why should one-time write-offs of fixed capital be used in absorption cost systems?
-
The table shows the number of people living in a house and the weight of trash (in pounds) at the curb just before trash pickup. People Trash (pounds) 2 ...........18 3 ...........33 6 ...........93...
-
With reference to Exercise 9.12, what randomized strategy will minimize Ms. Coopers maximum expected cost? In exercise Ms. Cooper is planning to attend a convention in Honolulu, and she must send in...
-
Assume that the current measurements in a strip of wire follow a normal distribution with a mean of 10 milliamperes and a variance of 4 (milliamperes) \({ }^{2}\). What is the probability that a...
-
Here is financial information for Pauletti Inc. InstructionsPrepare a schedule showing a horizontal analysis for 2012, using 2011 as the base year. December 31, 2012 December 31, 2011 Current assets...
-
Units Beginning work in process inventory 32,000 Percent Complete 65% Percent Complete 35% Units started this period 410,000 Completed and transferred out 412,000 Ending work in process inventory...
-
A bank has $650,000 in assets to allocate among investments in bonds, home mortgages, car loans, and personal loans. Bonds are expected to produce a return of 10%, mortgages 8.5%, car loans 9.5%, and...
-
Can anyone explain how analyzing variances from standard costs assist in identifying areas of operational improvement or cost reduction?
-
The cost of owning and running a home has fallen by nearly a fifth during the past year to its lowest level since 2006. The average property cost 7,298 a year to run in the 12 months to April, 17 per...
-
A lottery offers a \(\$ 1,000,000\) prize to be paid in 29 equal annual installments of \(\$ 20,000\) with a 30th final payment of \(\$ 420,000\). What is the total value of this annuity if the...
-
Select a car of your choice, find the list price, and calculate 5% and 10% price offers. Check out available money sources in your community, and prepare a report showing the different costs for the...
-
In Problems 20-52: a. State the type; and \(\mathbf{b}\). Answer the question. You deposit \(\$ 300\) at the end of each year into an account paying \(2 \%\) compounded annually. How much is in the...
-
Conduct a survey of banks, savings and loan companies, and credit unions in your area. Prepare a report on the different types of savings accounts available and the interest rates they pay. Include...
-
A gas (1.3, ho = 0.287 kJ/kgK) at a Mach number of 1.8, pressure of 0.8 bar and temperature of 373 K passes through a normal shock. Determine the density of the after the shock. gas
-
Do the three planes x + 2x + x 3 = 4, X X 3 = 1, and x + 3x = 0 have at least one common point of intersection? Explain.
-
Carbon dioxide (CO2) is the gas that is mainly responsible for global warming (the greenhouse effect). The burning of fossil fuels is a major cause of the increased concentration of CO2 in the...
-
Explain why the bond order of N2 is greater than that of N2+, but the bond order of O2 is less than that of O2+?
-
Calculate the pH and percent ionization of a 0.80 M HNO2 solution.
-
Suppose a job enrichment effort in one plant of a manufacturing firm is judged to be very successful. You are the corporate change agent responsible for the project, and you wish to diffuse it to...
-
Describe an example of resistance to change that you have observed. Why did it occur?
-
What qualities would the ideal gatekeeper possess to facilitate the communication of technical information in the firm?

Study smarter with the SolutionInn App


