Which of the following carbanion is least stable ? CH (1) (3) CH CH OCH3...
Fantastic news! We've Found the answer you've been seeking!
Question:
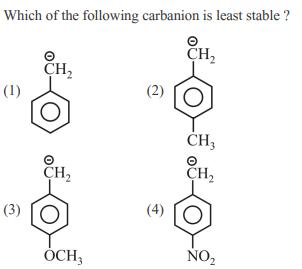
Transcribed Image Text:
Which of the following carbanion is least stable ? Ⓒ CH₂ (1) (3) CH₂ CH₂ OCH3 (2) CH3 O CH₂ NO₂ Which of the following carbanion is least stable ? Ⓒ CH₂ (1) (3) CH₂ CH₂ OCH3 (2) CH3 O CH₂ NO₂
Expert Answer:
Answer rating: 100% (QA)
As we see from all the options benzene ring is comm... View the full answer

Related Book For 

Posted Date:
Students also viewed these medical sciences questions
-
Laker Company reported the following January purchases and sales data for its only product. For specific identification, ending inventory consists of 2 7 0 units from the January 3 0 purchase, 5...
-
Q-Corporation uses the standard cost system and reports this years Actual Budget and Static Budget below. Total Assets: $24,975,500. Anticipated ROI was to be 17%. Budgeted sales were based on...
-
From a list of 468 small 2-year colleges a simple random sample of 100 colleges was drawn. The sample contained 54 public and 46 private colleges. Data for number of students (y) and number of...
-
Day and Night formed an accounting partnership in 2014. Capital transactions for Day and Night during 2014 are as follows: Partnership net income for the year ended December 31, 2014; is $68,400...
-
It is desired to produce an iron-carbon alloy that has a minimum hardness of 200 HB and a minimum ductility of 35% RA. Is such an alloy possible? If so, what will be its composition and...
-
1. For a monopolist, marginal revenue is ___________ (greater/less) than price. 2. A monopoly that cuts its price gains revenue from its ________customers but loses revenue from its...
-
The following function plays an important role in the experimental modal analysis: a. time-response function b. modal-response function c. frequency-response function
-
Colerain Corporation is a merchandising company that is preparing a profit plan for the third quarter of the calendar year. The companys balance sheet as of June 30 is shown below: Colerain...
-
A tensile strip of polystyrene that is 10 cm in length, 5 cm inwidth, and 2 cm in thickness is stretched to a length of 10.5 cm.Assuming that the sample is isotropic and deforms uniformly,calculate...
-
Landover Corporation is looking for a larger office building to house its expanding operations. It is considering two alternatives. The first is a newly constructed building at a cost of $6 million....
-
One of the most common challenges of managing social media in sports is finding the right balance between authenticity and professionalism. On one hand, athletes and teams want to be relatable and...
-
a. If Jim changes the quantities that he buys, will he buy more or fewer muffins and more or less coffee? b. When the prices change, will there be an income effect, a substitution effect, or both at...
-
Under IFRS, unrealized gains on non-trading stock investments should: (a) be reported as other revenues and gains in the income statement as part of net income. (b) be reported as other gains on the...
-
Under IFRS, at the end of the first year of operations, the total cost of the trading investments portfolio is $120,000. Total fair value is $115,000. The financial statement should show: (a) a...
-
The Wellstone Division operates as a profit center. It reports the following for the year. Budget Actual Sales $2,000,000 $1,860,000 Variable costs 800,000 760,000 Controllable fixed costs 550,000...
-
The father of scientific management is ____________. (a) Weber (b) Taylor (c) Mintzberg (d) Katz
-
Using Compary As calling plan, the cost of an everyeas phore call is a $1.05 cornection fee plus 19 ceris per minute. If the tatal cost of the call is 59.48, how lone is the phone call?
-
How has the too-big-to-fail policy been limited in the FDICIA legislation? How might limiting the too-big-to-fail policy help reduce the risk of a future banking crisis?
-
For each of these ions, draw the important resonance forms and predict which resonance form is likely to be the major contributor. (a) (b) (c) CH2 CH CH2 HN
-
Part of a synthesis by E. J. Corey and David Watt (Harvard University) involves the Diels-Alder cycloaddition of the following pyrone and cyclohexenone. The initial reaction gives the endo product,...
-
The compound BD3 is a deuterated form of borane. Predict the product formed when 1-methylcyclohexene reacts with BD3 THF ,followed by basic hydrogen peroxide.
-
A stock's beta is a measure of the stock's: A. total risk. B. market risk. C. unsystematic risk.
-
The larger the standard deviation of an asset's returns, the greater is the assets: A. total risk. B. market risk. C. unsystematic risk.
-
Which of the following is most likely considered a negative covenant? A. The company must maintain a current ratio of 2.0 or above. B. The company must maintain insurance on specific property. C. The...

Study smarter with the SolutionInn App


