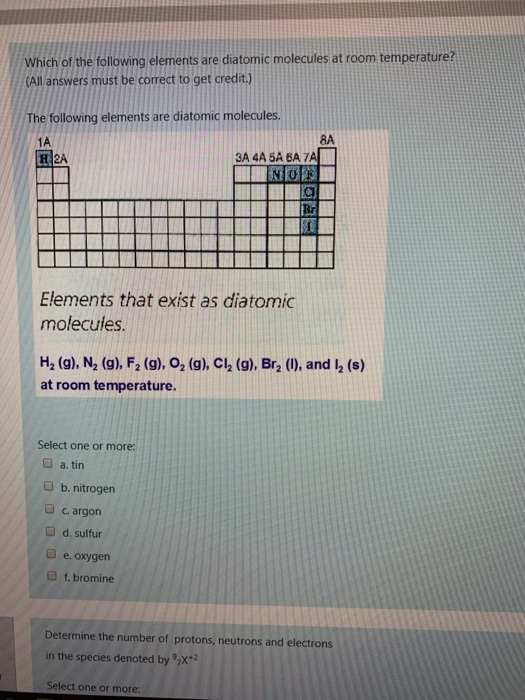
Which of the following elements are diatomic molecules at room temperature? (All answers must be correct...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
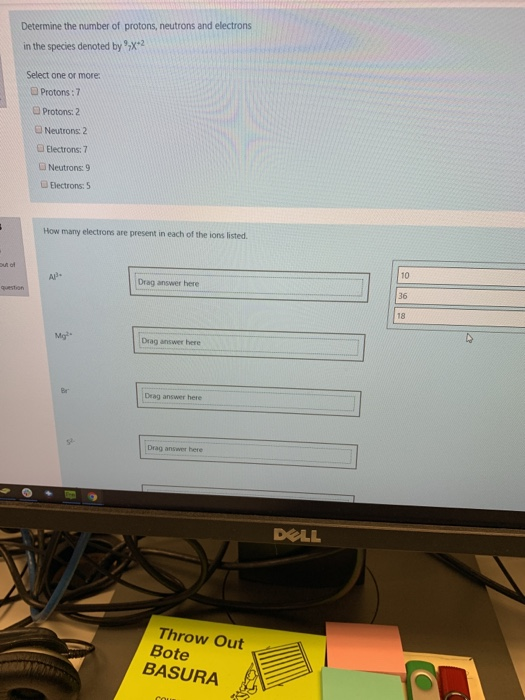
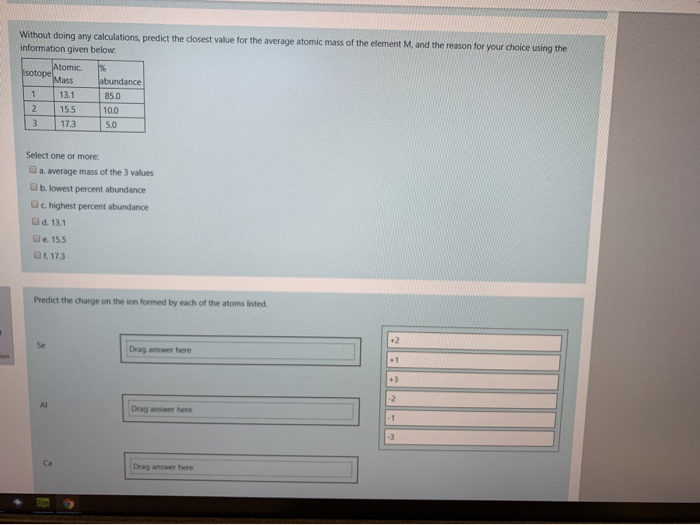
Which of the following elements are diatomic molecules at room temperature? (All answers must be correct to get credit.) The following elements are diatomic molecules. 1A H2A Elements that exist as diatomic molecules. Select one or more: a. tin b. nitrogen c. argon d. sulfur 3A 4A 5A 6A 7A NOF e. oxygen f. bromine H₂ (g), N₂ (g), F₂ (g), O₂ (g), Cl₂ (g), Br₂ (1), and I2₂ (s) at room temperature. C Br Select one or more: 8A Determine the number of protons, neutrons and electrons in the species denoted by X+2 S out of Determine the number of protons, neutrons and electrons in the species denoted by X-2 question Select one or more: Protons: 7 Protons: 2 Neutrons: 2 Electrons: 7 Neutrons: 9 Electrons: 5 How many electrons are present in each of the ions listed. Al- Mg Drag answer here Drag answer here Drag answer here Drag answer here Throw Out Bote BASURA COUR DELL 10 36 18 mon Without doing any calculations, predict the closest value for the average atomic mass of the element M, and the reason for your choice using the information given below. % sotope 1 2 3 Atomic. Mass 13.1 15.5 17.3 Select one or more: a. average mass of the 3 values b. lowest percent abundance c. highest percent abundance d. 13.1 e. 15.5 f. 17.3 Al 2 abundance 85.0 10.0 5.0 Predict the charge on the ion formed by each of the atoms listed. Ca Drag answer here Drag answer here Drag answer here +2 +1 +3 -2 -1 Which of the following elements are diatomic molecules at room temperature? (All answers must be correct to get credit.) The following elements are diatomic molecules. 1A H2A Elements that exist as diatomic molecules. Select one or more: a. tin b. nitrogen c. argon d. sulfur 3A 4A 5A 6A 7A NOF e. oxygen f. bromine H₂ (g), N₂ (g), F₂ (g), O₂ (g), Cl₂ (g), Br₂ (1), and I2₂ (s) at room temperature. C Br Select one or more: 8A Determine the number of protons, neutrons and electrons in the species denoted by X+2 S out of Determine the number of protons, neutrons and electrons in the species denoted by X-2 question Select one or more: Protons: 7 Protons: 2 Neutrons: 2 Electrons: 7 Neutrons: 9 Electrons: 5 How many electrons are present in each of the ions listed. Al- Mg Drag answer here Drag answer here Drag answer here Drag answer here Throw Out Bote BASURA COUR DELL 10 36 18 mon Without doing any calculations, predict the closest value for the average atomic mass of the element M, and the reason for your choice using the information given below. % sotope 1 2 3 Atomic. Mass 13.1 15.5 17.3 Select one or more: a. average mass of the 3 values b. lowest percent abundance c. highest percent abundance d. 13.1 e. 15.5 f. 17.3 Al 2 abundance 85.0 10.0 5.0 Predict the charge on the ion formed by each of the atoms listed. Ca Drag answer here Drag answer here Drag answer here +2 +1 +3 -2 -1
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which of the following molecules may show a pure rotational microwave absorption spectrum? (a) H20, (b) H202, (c) NH4 (d) N20?
-
Which of the following molecules may show a pure rotational Raman spectrum? (a) CH2Cl2 (b) CH3CH3, (c) SP6, (d) N2O?
-
Which of the following molecules may show infrared absorption spectra? (a) CH3CH3, (b) CH4, (c) CH3CI, (d) N2
-
Miami Tropical stock has a beta of 1.25. The risk-free rate of return is 4.34 percent and the market risk premium is 9.02 percent. What is the expected rate of return on this stock? 10.23 percent...
-
Multiple Choice Questions 1. In the bottom-up approach to cost estimating: (a) Required price is an input variable. (b) Cost estimates are an output variable. (c) Required price is an output...
-
Integrate by the first method or state why it does not apply and use the second method. Show the details. Re z dz, C the parabola y = 1+ (x 1)2 from 1+ i to 3 + 3i
-
Jack and Judy Myers are opening Parties on Demand. To buy stage props and other equipment they need outside capital, so they plan to organize the business as a corporation. They come to you for...
-
If a computer system does not have relocation hardware yet it implements swapping, then the memory manager will have to use a loader to recomputed the executable image from the absolute image. Would...
-
Sarah's Confectionery Company is a small manufacturer and wholesaler of lollipops, jellybabies and other sugary snacks.Sarah's Confectionery produces around 70% of the confectionary they sell, with...
-
From the adjustments columns in Exercise 5-9A, journalize the four adjusting entries, as of December 31, in proper general journal format. Exercise 5-9A Jim Jacobs Furniture Repair Work Sheet...
-
Jack Richkid is a 46-year-old accountant who is the Trustee of the Richkid Discretionary Trust whose beneficiaries include himself, his partner Jill (a 44-year-old voluntary social worker who did not...
-
Since products are very similar across the whole online-gifting industry? How could Flowaura differentiate itself as unique or a better choice for customers? How can the brand reduce the number of...
-
4. (10 points) Describe these sets by listing all members in these sets: a) S6 = {xe Nx>0^x 20} b) S7 = {x S6lx is prime} c) S8 = {xE S7|x is odd} d) Sg Zn (Z+UZ) = e) S10 = Sgn Sg
-
Explain the distinctions between single collections and embedded documents. Give an example of when utilizing a single collection is preferable to using embedded documents. Give an example of when...
-
Walmart is the largest retailer in the world with overall sales surpassing $ 5 0 0 billion. In fact, Walmart is still more than 3 x the size of Amazon Retail ( ~$ 1 6 3 billion ) in terms of overall...
-
Harris Company manufactures and sells a single product. A partially completed schedule of the company's total costs and costs per unit over the relevant range of 59,000 to 99,000 units is given...
-
Process of inserting an element in stack is called _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ Your answer: Pop Create Evaluation Enqueue Push Dequeue
-
Use integration by parts to evaluate the following. Check your answer by taking the derivative. x2e-xdx
-
The radius of a uranium-235 nucleus is about 7.0 10-3 pm. Calculate the density of the nucleus in g/cm3. (Assume the atomic mass is 235 amu.)
-
Calculate the total number of electrons that can occupy (a) One s orbital, (b) Three p orbitals, (c) Five dorbitals, (d) Seven forbitals.
-
The fluorides of the second-period elements and their melting points are: LiF, 845C; BeF2, 800C; BF3, 2126.7C; CF4, -184C; NF3, 2206.6C; OF2, -223.8C; F2, -219.6C. Classify the type(s) of...
-
The shaft consists of a solid segment \(A B\) and a hollow segment \(B C\), which are rigidly joined by the coupling at \(B\). If the shaft is made from A-36 steel, determine the maximum torque \(T\)...
-
The state of plane stress at a critical point in a steel machine bracket is shown. If the yield stress for steel is \(\sigma_{Y}=36 \mathrm{ksi}\), determine if yielding occurs using the maximum...
-
If \(\sigma_{Y}=50 \mathrm{ksi}\), determine the factor of safety for this state of stress against yielding, based on (a) the maximum shear stress theory and (b) the maximum distortion energy theory....

Study smarter with the SolutionInn App


