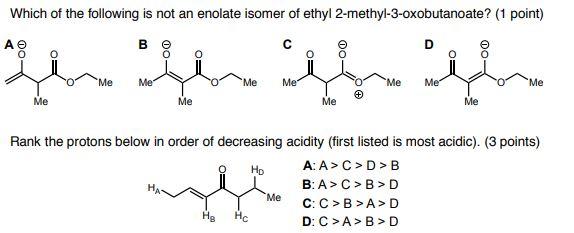
Which of the following is not an enolate isomer of ethyl 2-methyl-3-oxobutanoate? (1 point) ...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Which of the following is not an enolate isomer of ethyl 2-methyl-3-oxobutanoate? (1 point) سان من سلسة AⓇ Me Me B Q Me Me Me He He Me Me Me + Me Me Me Rank the protons below in order of decreasing acidity (first listed is most acidic). (3 points) AA> C > D>B Ho B: A> C >B> D C: C> B>A> D D: C>A> B> D Me Which of the following is not an enolate isomer of ethyl 2-methyl-3-oxobutanoate? (1 point) سان من سلسة AⓇ Me Me B Q Me Me Me He He Me Me Me + Me Me Me Rank the protons below in order of decreasing acidity (first listed is most acidic). (3 points) AA> C > D>B Ho B: A> C >B> D C: C> B>A> D D: C>A> B> D Me
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided bel... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Which of the following is not an internal audit objective designed to add value to a purchas-ing department? a. A review of the bidding process indicates that a vendor company may be operating under...
-
Which of the following is not an element: hydrogen, carbon, oxygen, water?
-
Which of the following is not an indicator that operating activities cash flows might be increased through distortion or manipulation? A. A significantly large increase in accounts payable. B. A...
-
Find all conjugates in C of the given number over the given field. 1+2 over Q
-
The Mendoza Company discussed in the chapter is now considering replacing a piece of equipment that the company uses to monitor the integrity of metal pipes used for deep-sea drilling purposes. The...
-
Use Laplace transforms to solve the initial value problems in Problems 1 through 16. x" + x = sin 2t; x (0) = 0 = x' (0)
-
Selected accounts and a section of a worksheet for Sunhill Stores are shown below. Required (a) The beginning and ending inventory were \($45\) 760 and \($52\) 420 respectively. Enter the beginning...
-
1. Explain how Googles strategy has been developed over the years. 2. What are the strengths and weaknesses of its approach? 3. In what ways should Googles approach to strategy development change in...
-
6. An annular disk of mass M, inner radius a and outer radius b is placed on a horizontal surface with coefficient of friction , as shown in the figure. At some time, an impulse Jox is applied at a...
-
Mr. M has been employed as an engineer by A Ltd., a company incorporated in Hong Kong. During the year ended 31 March 2019, Mr. M had the following income and expenditure. A monthly salary of...
-
The following selected transactions for Carla Vista company are presented in journal form. J1 Date Account Titles and Explanation Ref. Debit Credit May 5 Accounts Receivable 5,030 Service Revenue...
-
Although it is probably a clich to say we are living in a time of unprecedented technological change, it is difficult to truly comprehend the enormous changes that have taken place in a very short...
-
Verma Enterprises leases property to Pandey, Inc. Because Pandey, Inc. is experiencing financial difficulty, Verma agrees to receive five rents of 20,000 at the end of each year, with the rents...
-
The Wangs are planning for a retirement home. They estimate they will need 20,000,000 4 years from now to purchase this home. Assuming an interest rate of 10%, what amount must be deposited at the...
-
In its December 31, 2015, statement of financial position, Oakley Corporation reported as an asset, Net notes and accounts receivable, 7,100,000. What other disclosures are necessary?
-
How does an asset gain or loss develop in pension accounting?
-
7. What is the value of the following cash flows in year 6? What is the value today? Assume interest rates today are 6.5%, then rates will be 6.25% for the next 4 years, 3.75% for the next 3 years,...
-
For the next several days, take notes on your listening performance during at least a half-dozen situations in class, during social activities, and at work, if applicable. Referring to the traits of...
-
Division s of the trachea that enter lungs?
-
Why does diabetes result in high levels of glucose in the urine?
-
What is true of a solution that goes from pH 5 to pH 8? a. The H+ concentration decreases as the solution becomes more basic. b. The H+ concentration increases as the solution becomes more acidic. c....
-
The group \(\mathrm{D}_{3}\) in Schoenflies notation (32 in international notation, which is read "three-two"; see Table 5.1 ) consists of the proper (those not reflections or inversions) covering...
-
Derive the two-dimensional matrix representation Tic)=(2) Tin)=(3) Tex)=(37) (69) T(c2b)= 1 TO)-(71) 10-(11) TO=(9) = for the group D3, using the basis (e1, e2) defined in the following figure.
-
Prove that the matrix representation of \(\mathrm{D}_{3}\) worked out in Problem 5.6 is irreducible. Data from Problem 5.6 Derive the two-dimensional matrix representation Tic)=(2) Tin)=(3)...

Study smarter with the SolutionInn App


