Which of the following lines correctly show the temperature dependence of equilibrium constant, K, for an...
Fantastic news! We've Found the answer you've been seeking!
Question:
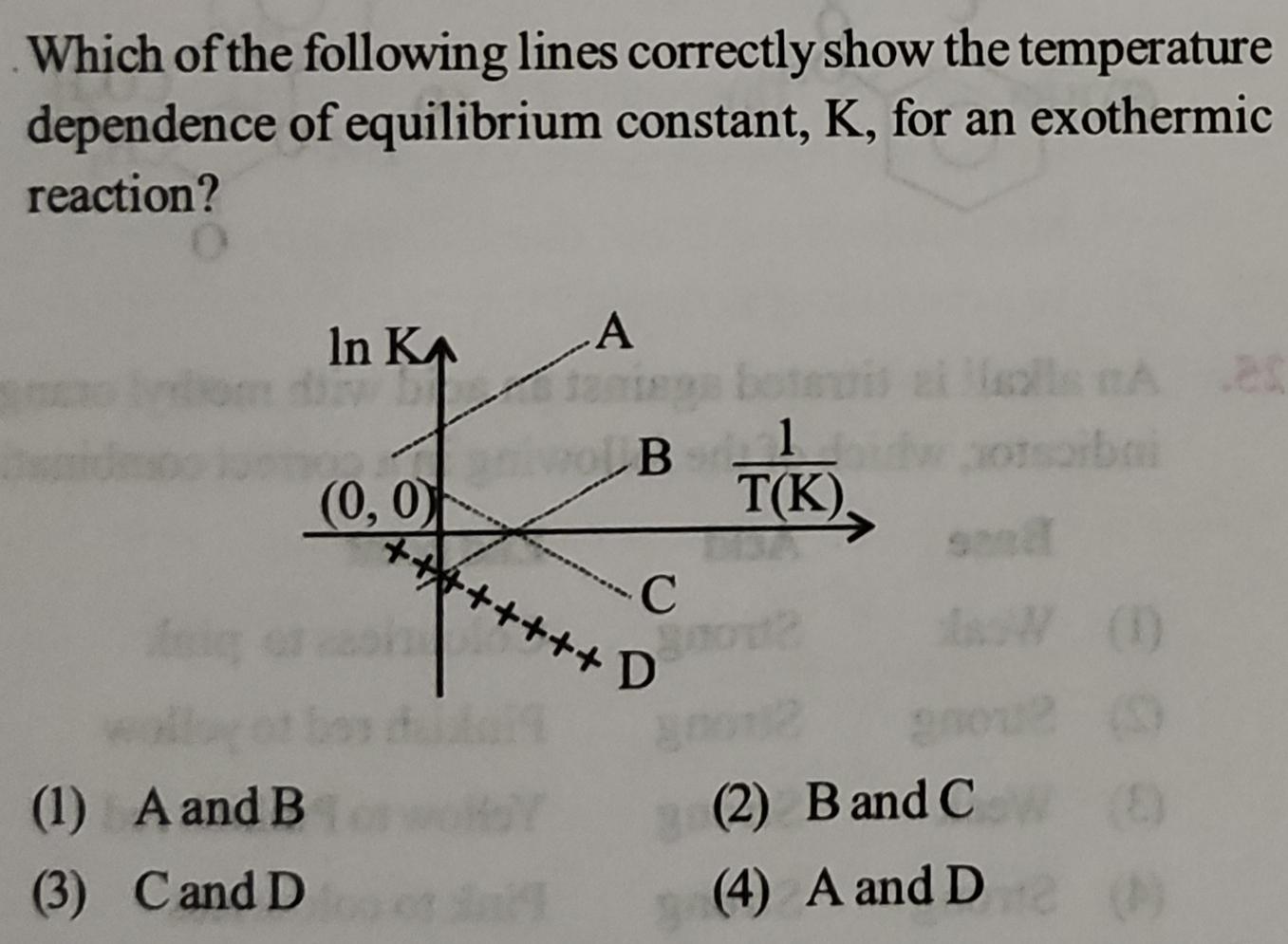
Transcribed Image Text:
Which of the following lines correctly show the temperature dependence of equilibrium constant, K, for an exothermic reaction? (1) A and B (2) B and C (3) C and D (4) A and D Which of the following lines correctly show the temperature dependence of equilibrium constant, K, for an exothermic reaction? (1) A and B (2) B and C (3) C and D (4) A and D
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which of the following 2 x 2 matrices are positive definite? (a) (b) (c) (d) (e) (f) In the positive definite cases, write down the formula for the associated inner product. 0 2 2 1 2 2 3 (1 -3)
-
Which of the following C corporations will be allowed to use the cash method of accounting for 2017? Explain your answers. a. Jade Corporation, which had gross receipts of $5.3 million in 2014, $4.1...
-
Which of the following compounds show only a single peak in their 1H NMR spectrum? a. CH3CH2OCH2CH3 b. c. CH,CH,CCI
-
With reference to the Auditors Report, answer the following questions: The recent final audit report of an FMCG carries a line which is read as in the manner so required and respectively give a true...
-
ELV Trade Mart has recently had lackluster sales. The rate of inventory turnover has dropped, and the merchandise is gathering dust. At the same time, competition has forced ELVs suppliers to lower...
-
This case is based on Under Armour, Inc.s, consolidated balance sheets, consolidated statements of income, and Note 2 of its financial statements (Significant Accounting Policies) found online in the...
-
How long does it take an ambulance to respond to a request for emergency medical aid? One of the goals of one study was to estimate the response time of ambulances using warning lights (Ho \&...
-
1. What options are available to Nils? 2. What would you do and why? On December 26, 2004, an earthquake reaching 9.1 on the Richter scale triggered a series of devastating tsunamis off the coast of...
-
Green Trifle Bakery discovered some errors for 2020 and 2021. What would be the impact on net income for each year if the errors were left undetected? Error Ending inventory in 2020 2020 2021 was...
-
BookWeb, Inc., sells books and software over the Internet. A recent article in a trade journal has caught the attention of management because the company has experienced soaring inventory handling...
-
This week we are learning about financial statement analysis and how to compute various ratios. This is a great way to understand how a company is doing. However, what are some limitations of...
-
Sequential consistency (SC) requires that all reads and writes appear to have executed in some total order. This may require the processor to stall in certain cases before committing a read or write...
-
It is critical that the scoreboard be able to distinguish RAW and WAR hazards, because a WAR hazard requires stalling the instruction doing the writing until the instruction reading an operand...
-
You are building a system around a processor with in-order execution that runs at 1.1 GHz and has a CPI of 1.35 excluding memory accesses. The only instructions that read or write data from memory...
-
Find three run-time errors in the following program. public class Has Errors { } public static void main(String[] args) { } int x = 0; int y = 0; Scanner in new Scanner("System.in");...
-
Propose one possible way to improve TCO by focusing on reducing server power. What are the challenges to evaluating your proposal? Estimate the TCO improvements based on your proposal. What are some...
-
Write each vector as a linear combination of the vectors in S. (Use 5 and 52, respectively, for the vectors in the set. If not possible, enter IMPOSSIBLE.) S = {(1, 2, 2), (2, -1, 1)} (a) z = (-9,...
-
APC16550D UART has a clock running at18.432 MHz and its baud rate is set to 2000.Determine the HEX contents of its DLM and DLL registers. Please can you explain step by step and in detail how you get...
-
The atoms in a single bond can rotate about the internuclear axis without breaking the bond. The atoms in a double bond and a triple bond cannot rotate about the internuclear axis unless the bond is...
-
In the flushing and cleaning of columns used in liquid chromatography, a series of solvents is used. Hexane (C6H14), chloroform (CHCl3), methanol (CH3OH), and water are passed through the column in...
-
Ethanethiol (C2H5SH; also called ethyl mercaptan) is commonly added to natural gas to provide the rotten egg smell of a gas leak. The boiling point of ethanethiol is 35C and its heat of vaporization...
-
Study Figure 13.6. Write a short report on Bayers management of its cash flows over the period reported. Figure 13.6 Bayer Group's consolidated statement of cash flows Income after taxes Income taxes...
-
Expenses and revenues are subjective; cash flows are facts. Therefore cash flow statements cannot mislead. Discuss.
-
Repeat Exercise 13.3, but this time work on the assumption that non-current assets that had originally cost :30,000, with accumulated depreciation of :12,000, had been sold during the year ended 31...

Study smarter with the SolutionInn App



