Which of the following represents the correct balanced equation for the dissolution reaction of Na3PO4 (S)?...
Fantastic news! We've Found the answer you've been seeking!
Question:



Transcribed Image Text:
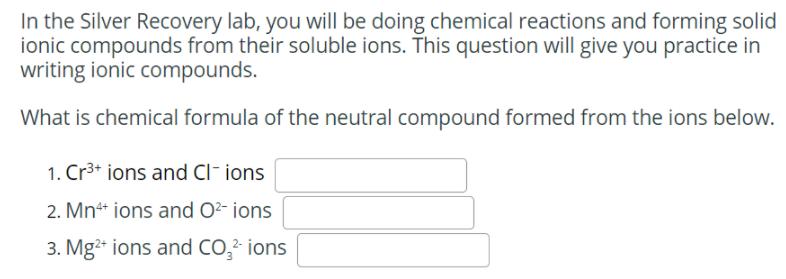
Which of the following represents the correct balanced equation for the dissolution reaction of Na3PO4 (S)? O NazPO4 (s) → 3 Na* (aq) + 4 PO4 (aq) 1- NazPO4 (s) → 3 Na3+ (aq) + 3 PO41- (aq) O NazPO4 (s) – Na3+ (aq) + PO43 (aq) NazPO4 (s) → 3 Na* (aq) + PO43 (aq) In the Silver Recovery lab, you will be doing chemical reactions and forming solid ionic compounds from their soluble ions. This question will give you practice in writing ionic compounds. What is chemical formula of the neutral compound formed from the ions below. 1. Cr3+ ions and Cl- ions 2. Mn+ ions and O- ions 3. Mg ions and CO,? ions There are two possible methods for recovering copper from a solution of copper (II) sulfate, one uses precipitation and the other uses redox. Copper(II) ions can be precipitated as copper(II) carbonate. Cuso,(aq) + Na,CO;(aq) → CuCO3(s) + NazSO4(aq) How many mL of 0.422 M Na2CO3 would be needed to precipitate all of the copper ions in 24.5 mL of 0.508 M CuSO4? mL How many grams of CuCO3(s) would be formed from the copper ions in 24.5 mL of 0.508 M CuSO4? Copper(II) ions can be displaced by aluminum metal. 3 CusO4(aq) + 2 Al(s) → Al,(SO4)3(aq) + 3 Cu(s) How many grams of Al would be needed to displace all of the copper ions in 58.7 mL of 0.740 M CuSO4? How many grams of Cu would be recovered from the copper ions in 58.7 mL of 0.740 M CuSO4? Which of the following represents the correct balanced equation for the dissolution reaction of Na3PO4 (S)? O NazPO4 (s) → 3 Na* (aq) + 4 PO4 (aq) 1- NazPO4 (s) → 3 Na3+ (aq) + 3 PO41- (aq) O NazPO4 (s) – Na3+ (aq) + PO43 (aq) NazPO4 (s) → 3 Na* (aq) + PO43 (aq) In the Silver Recovery lab, you will be doing chemical reactions and forming solid ionic compounds from their soluble ions. This question will give you practice in writing ionic compounds. What is chemical formula of the neutral compound formed from the ions below. 1. Cr3+ ions and Cl- ions 2. Mn+ ions and O- ions 3. Mg ions and CO,? ions There are two possible methods for recovering copper from a solution of copper (II) sulfate, one uses precipitation and the other uses redox. Copper(II) ions can be precipitated as copper(II) carbonate. Cuso,(aq) + Na,CO;(aq) → CuCO3(s) + NazSO4(aq) How many mL of 0.422 M Na2CO3 would be needed to precipitate all of the copper ions in 24.5 mL of 0.508 M CuSO4? mL How many grams of CuCO3(s) would be formed from the copper ions in 24.5 mL of 0.508 M CuSO4? Copper(II) ions can be displaced by aluminum metal. 3 CusO4(aq) + 2 Al(s) → Al,(SO4)3(aq) + 3 Cu(s) How many grams of Al would be needed to displace all of the copper ions in 58.7 mL of 0.740 M CuSO4? How many grams of Cu would be recovered from the copper ions in 58.7 mL of 0.740 M CuSO4?
Expert Answer:
Answer rating: 100% (QA)
Dissolution Reaction of NaPo cs Na Caq Pe Cap to s 1 Cra 36L Caq CrcL Cs 2 M Caq 20 94 2t Cu... View the full answer

Related Book For 

Fundamentals of Investments, Valuation and Management
ISBN: 978-1259720697
8th edition
Authors: Bradford Jordan, Thomas Miller, Steve Dolvin
Posted Date:
Students also viewed these accounting questions
-
Which of the following represents the best investment advice? a. Avoid Texas because its expected return is lower than its required return. b. Buy Montana and Texas because their required returns are...
-
Which of the following represents the maximun amortization period mandated for an intangible asset with finite useful life? a. 10 years b. 20 years c. 40 d. No arbitary cap on the useful life has...
-
Which one of the following represents the correct cause-and-effect sequence of leading and lagging measures in an organization? a. Learning and Growth S Internal Processes S Customer S Financial b....
-
Did you have imaginary friends when you were a young child? What do you remember about them and their value to you?
-
You want to change the mutual fund you have your IRA invested in. How might you change funds without triggering a tax on your withdrawal?
-
In April 2022 Lucas Arcand opened a new cleaning service in Victoria. Please assist him by journalizing the following business transactions: 2022 Apr. 1 Lucas Arcand invested $6,000 worth of cleaning...
-
The [+45/-45] laminate described in Problem 7.6 is subjected to a uniaxial force per unit length \(N_{x}=30 \mathrm{MPa} \mathrm{mm}\). Find the resulting stresses and strains in each ply along the...
-
Squirrel Tree Services reports the following amounts on December 31. In addition, the company reported the following cash flows. Required: 1. Prepare a balance sheet. 2. Prepare a statement of...
-
In your own opinion, what makes the filed of Applied Behavior Analysis different from other approaches to understanding and changing human behavior?
-
Palmer, a U.S. company, acquired 90% of Scalas voting stock for $32,600 in cash on January 1, 2019, when Scalas book value was $5,000. The fair value of the noncontrolling interest at the date of...
-
Example: How many numbers 12 L357 2. Write a C++ code using Loops to find the summation of the first n odd numbers. Example is given below: 1 overw 11 The summation of the first 12 numbers=36
-
What is the output of the following? A. e B. d C. ed D. None of the above 1: public class Legos { 2: 3: 4: 5: 6: 7: 8: 9: } public static void main(String[] args) { var sb = new StringBuilder();...
-
Which is one of the lines output by this code? A. [] B. [8, 10] C. [8, 9, 10] D. [10, 8] E. The code does not compile. 10: var list= new ArrayList (); 11: list.add (10); 12: list.add (9); 13:...
-
What is the result of compiling and executing the following class? A. The code does not compile due to line x. B. The code does not compile due to line y. C. 31 D. 61 public class Tolls { private...
-
What is the output of the following class? A. 2 B. 3 C. 4 D. None of the above 1: package rocket; 2: public class Countdown { 3: 4: 5: 6: 7: 8: } public static void main(String[] args) { var builder...
-
Which of the following can fill in the blank so the code prints true? var happy :) ( "; var really happy.trim(); var question = = System.out.println(really.equals(question)); A. happy.substring(0, B....
-
Discuss price ceilings and price floors. The government sometimes imposes price controls on certain goods and services at certain times. However, there are usually unintended consequences. Discuss...
-
A new car sold for $31,000. If the vehicle loses 15% of its value each year, how much will it be worth after 10 years?
-
The Layton Growth Fund has an alpha of 2.1 percent. You have determined that Laytons information ratio is 0.5. What must Laytons tracking error be relative to its benchmark?
-
What is the dollar value of an 01 for the bond in Problem 23? Data From Problem 23 What is the Macaulay duration of a 7 percent coupon bond with five years to maturity and a current price of...
-
You have been given the following return information for two mutual funds (Papa and Mama), the market index, and the risk-free rate. Calculate the Sharpe ratio, Treynor ratio, Jensens alpha,...
-
You have a solenoid for which a current \(I\) per winding produces a magnetic field of magnitude \(B_{1}\) inside the soleniod. You cut the solenoid open parallel to its symmetry axis. You then...
-
You need to design a square toroid such that the magnetic field in the centre of the square windings is \(0.0020 \mathrm{~T}\), and the side length of a square winding is \(3 \mathrm{~cm}\). The wire...
-
The force, \(F\), of the wind blowing against a building is given by \(F=C_{D} ho V^{2} A / 2\), where \(V\) is the wind speed, \(ho\) the density of the air, \(A\) the cross-sectional area of the...
Still Breathing My Journey With Love Loss And Reinvention 1st Edition - ISBN: B0B2T4XWHH - Free Book

Study smarter with the SolutionInn App


