Which of the following statements about Lewis structures is FALSE? An octet is when an atom...
Fantastic news! We've Found the answer you've been seeking!
Question:

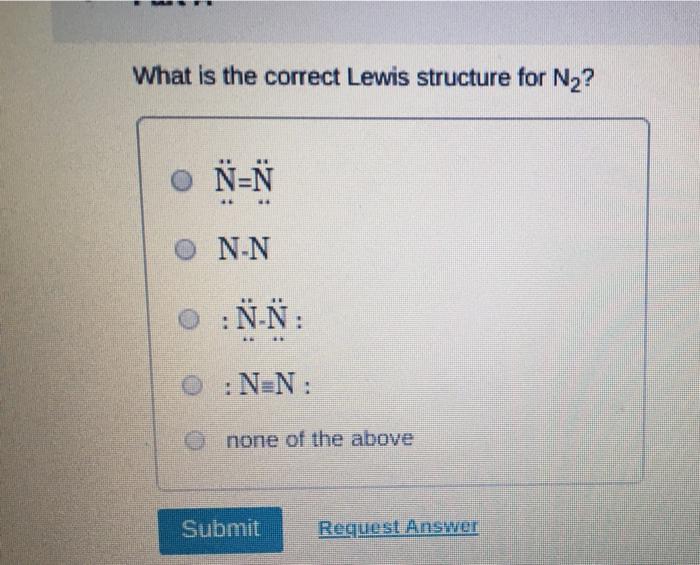
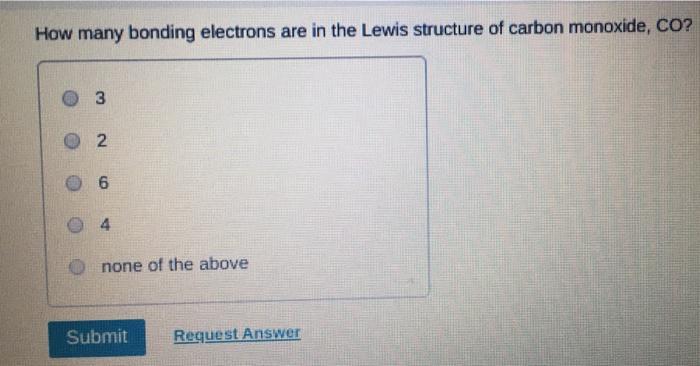

Transcribed Image Text:
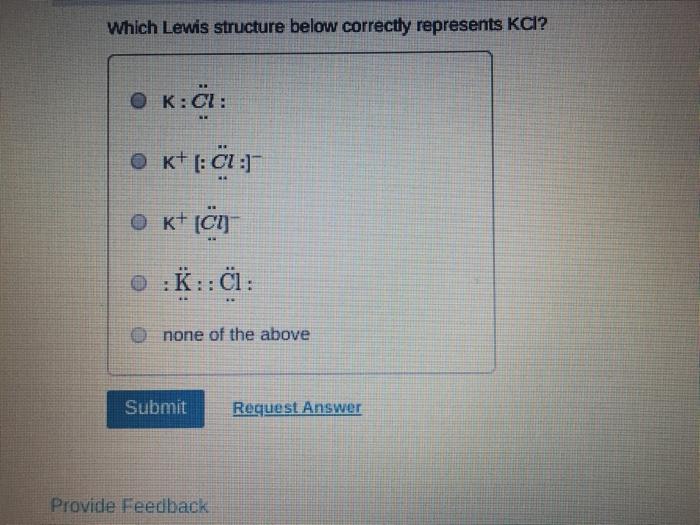
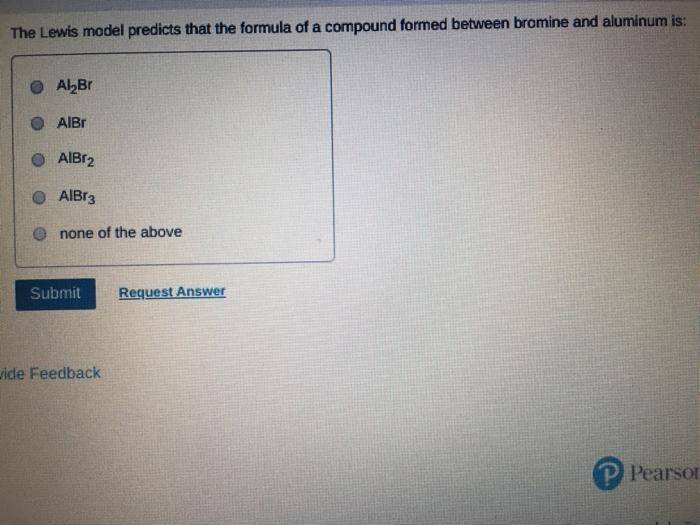
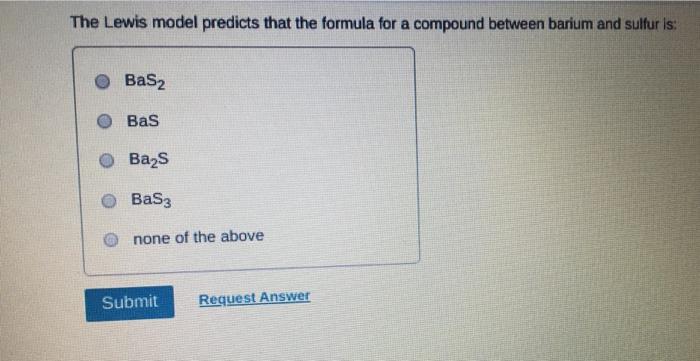
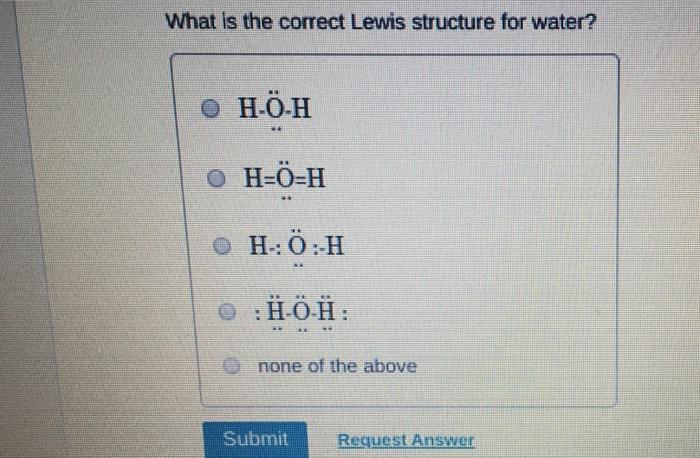
Which of the following statements about Lewis structures is FALSE? An octet is when an atom has 8 valence electrons. A covalent bond occurs when electrons are shared. An ionic bond occurs when electrons are transferred. ⒸA duet is a stable electron configuration for helium. All of the above statements are true. Submit Provide Feedback Request Answer Which Lewis structure below correctly represents KCI? K: Cl: ● K+ [: C:] ⒸK+ [CI] :K::C: none of the above Submit Request Answer Provide Feedback The Lewis model predicts that the formula of a compound formed between bromine and aluminum is: Al₂Br AlBr AlBr₂ 0 AlBr3 none of the above Submit Request Answer ide Feedback P Pearson The Lewis model predicts that the formula for a compound between barium and sulfur is: BaS₂ Bas Ba₂S BaS3 none of the above Submit Request Answer What is the correct Lewis structure for water? OH-Ö-H OH-Ö-H OH: Ö:-H © HOH: none of the above Submit Request Answer What is the correct Lewis structure for N₂? 0 N=N N-N : Ñ-Ñ : : N=N: none of the above Submit Request Answer How many bonding electrons are in the Lewis structure of carbon monoxide, CO? 3 2 6 none of the above Submit Request Answer 3- The total number of electrons to be counted for the Lewis structure of the PO4³ polyatomic ion is: 26. 29. 8. 32. none of the above Submit Request Answer Which of the following statements about Lewis structures is FALSE? An octet is when an atom has 8 valence electrons. A covalent bond occurs when electrons are shared. An ionic bond occurs when electrons are transferred. ⒸA duet is a stable electron configuration for helium. All of the above statements are true. Submit Provide Feedback Request Answer Which Lewis structure below correctly represents KCI? K: Cl: ● K+ [: C:] ⒸK+ [CI] :K::C: none of the above Submit Request Answer Provide Feedback The Lewis model predicts that the formula of a compound formed between bromine and aluminum is: Al₂Br AlBr AlBr₂ 0 AlBr3 none of the above Submit Request Answer ide Feedback P Pearson The Lewis model predicts that the formula for a compound between barium and sulfur is: BaS₂ Bas Ba₂S BaS3 none of the above Submit Request Answer What is the correct Lewis structure for water? OH-Ö-H OH-Ö-H OH: Ö:-H © HOH: none of the above Submit Request Answer What is the correct Lewis structure for N₂? 0 N=N N-N : Ñ-Ñ : : N=N: none of the above Submit Request Answer How many bonding electrons are in the Lewis structure of carbon monoxide, CO? 3 2 6 none of the above Submit Request Answer 3- The total number of electrons to be counted for the Lewis structure of the PO4³ polyatomic ion is: 26. 29. 8. 32. none of the above Submit Request Answer
Expert Answer:
Answer rating: 100% (QA)
1lewis structure is a very simplified representation of the valence shell electrons in a moleculeIt ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which of the following statements about the minimum variance portfolio of all risky securities are valid? (Assume short sales are allowed.) Explain. a. Its variance must be lower than those of all...
-
Which of the following statements about the security market line (SML) are true? a. The SML provides a benchmark for evaluating expected investment performance. b. The SML leads all investors to...
-
Which of the following statements about the project budgeting process is true? The weekly budget is formed by only using the early start times of each activity. The weekly budget is formed by only...
-
We-B-Busy, Inc. uses job costing and a predetermined overheadrate based onmachine hours (MH). The firm estimated that it would use $240,000of MOH and7,500 MH this year. Data for February:Job 1010 2...
-
Tiger Trade has the following cash transactions for the period. Accounts Amounts Cash received from sale of products to customers ...... $ 40,000 Cash received from the bank for long- term loan...
-
The Bijou Theater shows vintage movies. Customers arrive at the theater line at the rate of 100 per hour. The ticket seller averages 30 seconds per customer, which includes placing validation stamps...
-
Plaintiffs James and Betty Tonkovich own approximately 850 acres of in Belmont County, Ohio. Plaintiffs belong to a group of landowners known as Belmont Leasing Group, which leases land for oil and...
-
A mixture containing 65.0 mole% acetone (Ac) and the balance acetic acid (AA) is separated in a continuous distillation column at 1 atm. A flowchart for the operation is as follows: The overhead...
-
Path for Ampere's law Plane surface Bulging surface A parallel-plate capacitor with plates of radius R is being charged by the current ic. Recall Ampere's Law B.d-po Although there is no current...
-
When one hears the word entrepreneur, many faces come to mind. Elon Musk, Sara Blakely, Warren Buffet, Martha Stewart, Bill Gates, and Daymond John are just a few that might spring to mind. There are...
-
Which requirements address unique product and service test procedures and document the end-state customer experience? Organizational Requirements Technical Requirements Customer Requirements...
-
Please help me in solving this as per IFRS 1 5 ( 1 5 ( Also calculate annual service revenue with the discount factor ) ) Modern Engineering ( ( ME ) ) entered into a contract for 3 3 - year...
-
Kirpalani Stores manufactures customized household furnishings. The company uses a perpetual inventory system and has a highly labour-intensive production process, so it assigns manufacturing...
-
You are competing with your friends to type text messages on your smartphone as quickly as possible. Here are the rules: you use two thumbs for texting and they start out on the bottom left and...
-
What is the alpha of each stock? factor premium MKT-RF SMB HML UMD 0.65% 0.21% 0.39% 0.67% standard deviation of factor return 5.37% 3.21% 3.50% 4.74% Consider the stocks detailed in the below table....
-
If a person does not have to pay more than $3,000 in a year for health care expenses, this is called their maximum out-of-pocket. deductible co-payment lifetime maximum
-
Hudson, Inc. is a calendar year corporation. Its financial statements for the years 2015 and 2014 contained the following errors: 2015 2014 Ending inventory $6,000 overestimated $16,000 overestimated...
-
Apply Jacobis method to the given system. Take the zero vector as the initial approximation and work with four-significant-digit accuracy until two successive iterates agree within 0.001 in each...
-
The equilibrium constant is 0.0900 at 25oC for the reaction H2O(g) + Cl2O(g) 2HOCl(g) For which of the following sets of conditions is the system at equilibrium? For those which are not at...
-
Consider the reaction P4(g) 2P2(g) where Kp = 1.00 10-1 at 1325 K. In an experiment where P4(g) was placed in a container at 1325 K, the equilibrium mixture of P4(g) and P2(g) has a total pressure...
-
Three elements have the electron configurations 1s22s22p63s23p6, 1s22s22p63s2, and 1s22s22p63s23p64s1. The first ionization energies of these elements (not in the same order) are 0.419, 0.735, and...
-
How are risk and return each defined? Do you think there is a relationship between them?
-
Describe the components of a business plan. How does each of these components help the following people to make decisions about a business? (a) An investor. (b) A creditor. (c) A manager or owner.
-
Define relevant range. Why is it important?

Study smarter with the SolutionInn App


