Which one of the following values is the most likely value for the linear correlation coefficient...
Fantastic news! We've Found the answer you've been seeking!
Question:
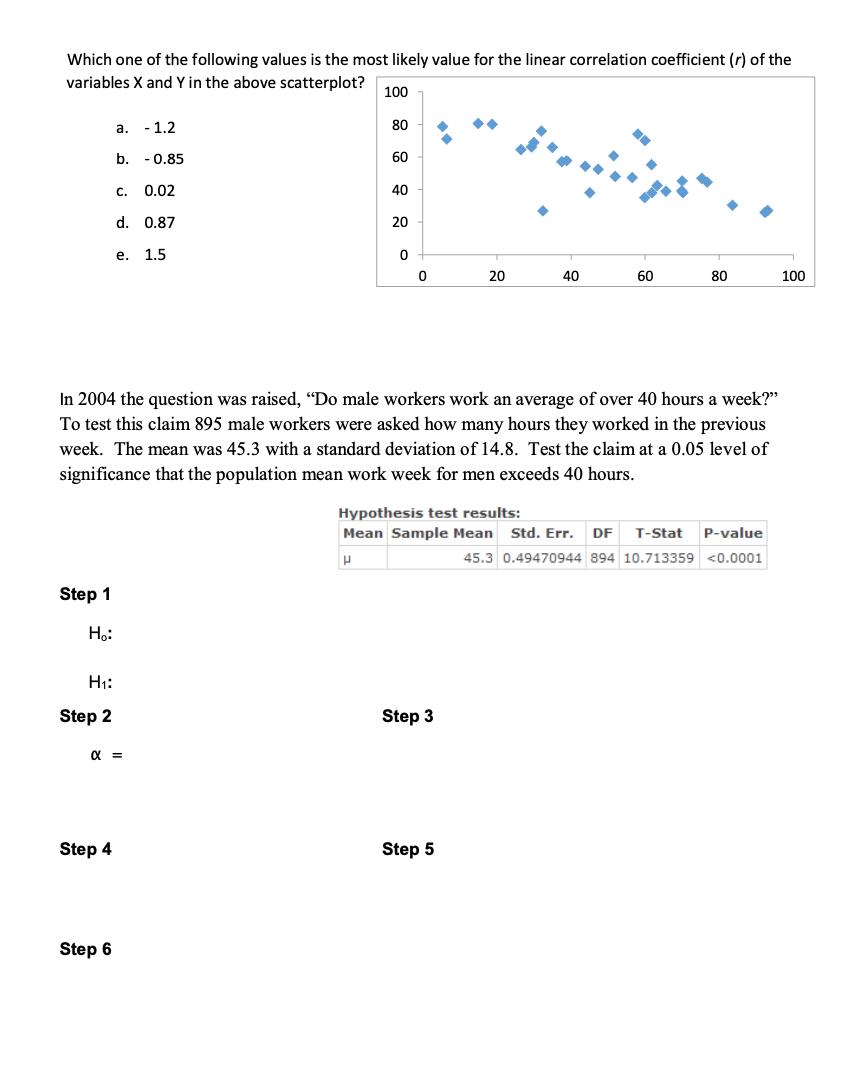
Transcribed Image Text:
Which one of the following values is the most likely value for the linear correlation coefficient (r) of the variables X and Y in the above scatterplot? Step 1 H₂: H₁: Step 2 a. - 1.2 b. Step 4 Step 6 C. 0.02 d. 0.87 e. 1.5 α = 0.85 100 80 60 40 20 0 0 In 2004 the question was raised, "Do male workers work an average of over 40 hours a week?" To test this claim 895 male workers were asked how many hours they worked in the previous week. The mean was 45.3 with a standard deviation of 14.8. Test the claim at a 0.05 level of significance that the population mean work week for men exceeds 40 hours. 20 Step 3 40 Step 5 60 80 Hypothesis test results: Mean Sample Mean Std. Err. DF T-Stat P-value P 45.3 0.49470944 894 10.713359 <0.0001 100 Which one of the following values is the most likely value for the linear correlation coefficient (r) of the variables X and Y in the above scatterplot? Step 1 H₂: H₁: Step 2 a. - 1.2 b. Step 4 Step 6 C. 0.02 d. 0.87 e. 1.5 α = 0.85 100 80 60 40 20 0 0 In 2004 the question was raised, "Do male workers work an average of over 40 hours a week?" To test this claim 895 male workers were asked how many hours they worked in the previous week. The mean was 45.3 with a standard deviation of 14.8. Test the claim at a 0.05 level of significance that the population mean work week for men exceeds 40 hours. 20 Step 3 40 Step 5 60 80 Hypothesis test results: Mean Sample Mean Std. Err. DF T-Stat P-value P 45.3 0.49470944 894 10.713359 <0.0001 100
Expert Answer:
Answer rating: 100% (QA)
Answer From the given Scatter plot Y 085 Given that X 453 S 148 2 005 n ... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these economics questions
-
Which one of the following compounds is most consistent with the infrared spectrum given in Figure 13.31? Explain your reasoning.
-
Which one of the following is correct? a. Management accounting reports are usually available to the public. b. Management accounting is legislated and governed by regulatory agencies. c. Financial...
-
Which one of the following is incorrect? a. Management accounting is not as important as financial accounting for the competitive success of a company. b. Governments do not require a company to...
-
Consider the maleic anhydride process shown in Appendix B.5. Estimate the profit margin for this process using the costs of raw materials and products from Table 8.4. Figure B.5.1 Table 8.4 Costs of...
-
Estimate the remaining life in revolutions of an 02-30 mm angular-contact ball bearing already subjected to 200 000 revolutions with a radial load of 18 kN, if it is now to be subjected to a change...
-
A customer account in the accounts receivable subsidiary ledger of Kettle Office Supplies follows: Required Describe the three posted transactions. 112590 Debit Balance 1,600 6,320 5,560 2,760 Beaver...
-
Define the two main environmental objectives proposed for management of companies: sustainable development and eco-efficiency. Are these objectives related?
-
On January 1, Year 6, HD Ltd., a building supply company, JC Ltd., a construction company, and Mr. Saeid, a private investor, signed an agreement to carry out a joint operation under the following...
-
Question 2 Suppose that the date when Lehman Brothers filed for bankruptcy is taken as the approximate start of the late 2000s financial crisis. Suppose also that you have two share prices for each...
-
The Chartered Financial Analyst (CFA) designation is the de facto professional certification for the financial industry. Employers encourage their prospective employees to complete the CFA exam....
-
Looking for a resource or if you can help me with type of retainer agreement best for following type of cases: Family Law (simple divorce, adoption) Personal injury (where client has no money but has...
-
Solve the system [4x-6y-z=11; 3x-6y= 15; 9x=27]
-
Consider a flow network G = (V,E) with positive edge capacities {c(e)}. Let f: E Ro be a maximum flow in G, and G be the residual graph. Denote by S the set of nodes reachable from s in G and by T...
-
A horizontal force of 55.0 N is required to pull an 10.0 kg block at a constant velocity across a horizontal wooden desk. Determine the coefficient of kinetic friction acting between the block and...
-
After five formal, Inc.. Is considering making an investment in a new cutting machine for its recently acquired Hollywood factory. The finance department is working on the analysis for the...
-
Manganese serves as a cofactor in enzyme-catalyzed reactions describe its catalytic role? If manganese is not permanently bound to an organic molecule is it bound transiently and how?
-
Consider the following decision makers (with w, indicating the wealth state and U indicating the utility function over wealth): Ben: Wo $50; U(w) = 4/w = Will: Wo $50; U(w) - w Liz: wo= $50; U(w)=5...
-
Distinguish among total-moisture content, free-moisture content, equilibrium-moisture content, unbound moisture, and bound moisture.
-
Sophie is a single taxpayer. For the first payroll period in October 2012, she is paid wages of $3,250 monthly. Sophie claims three allowances on her Form W-4. a. Use the percentage method to...
-
Frank, age 35, and Joyce, age 34, are married and file a joint income tax return for 2012. Their salaries for the year total $83,000 and they have taxable interest income of $4,000. They have no...
-
Yolanda is a cash basis taxpayer with the following transactions during the year: Cash received from sales of products........................................................................$65,000...
-
What is reaction coordinate? What is its significance in chemical reaction?
-
Show that for steady state flow process, \(\Delta H=Q-W_{S}\).
-
An ideal gas \(\left(C_{P}=5, C_{V}=3 ight)\) is changed from \(1 \mathrm{~atm}\) and \(22.4 \mathrm{~m}^{3}\) to \(10 \mathrm{~atm}\) and 2.24 \(\mathrm{m}^{3}\) by the reversible process of heating...

Study smarter with the SolutionInn App


