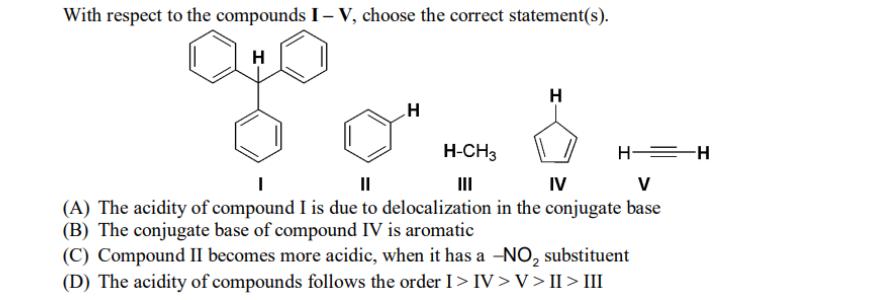
With respect to the compounds I-V, choose the correct statement(s). H H H-CH3 H H=H ||...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
With respect to the compounds I-V, choose the correct statement(s). H H H-CH3 H H=H || IV V (A) The acidity of compound I is due to delocalization in the conjugate base (B) The conjugate base of compound IV is aromatic (C) Compound II becomes more acidic, when it has a -NO₂ substituent (D) The acidity of compounds follows the order I > IV>V>II>III With respect to the compounds I-V, choose the correct statement(s). H H H-CH3 H H=H || IV V (A) The acidity of compound I is due to delocalization in the conjugate base (B) The conjugate base of compound IV is aromatic (C) Compound II becomes more acidic, when it has a -NO₂ substituent (D) The acidity of compounds follows the order I > IV>V>II>III
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The Hermite polynomials are orthogonal with respect to the inner product Find the first five monic Hermite polynomials. 00 (f. 8) = ! f(1)8(1) e-* dr. dt. J-00
-
Using the parallel-axis theorem, determine the product of inertia of the area shown with respect to the centroidal x and y axes. 16 in 892 0,61 in 16.5 in
-
Determine the mass moment of inertia of the steel machine element shown with respect to the z axis. (The density of steel is 7850 kg/m3.) 360 mm 80 mm 240 min 50 mm- 100 mm
-
In Problems 5978, solve each equation in the complex number system. x 2 + 25 = 0
-
Was the contract too unreasonable to enforce? Mason was a surgeon/apothecary in the English town of Thetford. Davis wished to apprentice himself to Mason. The two agreed that Davis would work for...
-
Transaction data for Royale Real Estate Agency are presented in E2.4. Instructions Journalize the transactions. E2.4 Oct. 1 James Royale begins business as a real estate agent with a cash investment...
-
Describe the differences between domestic and international HRM.
-
With gasoline prices increasing, drivers are more concerned with their cars gasoline consumption. For the past 5 years a driver has tracked the gas mileage of his car and found that the variance from...
-
Need to calculate payback period for each project Project A Year 0 - $-8,000,000 Year 1 - $4,000,000 Year 2 - $2,500,000 Year 3 - $2,500,000 Project B Year 0 - $-4,250,000 Year 1 - $1,500,000 Year 2...
-
Two-dimensional dynamics often involves solving for two unknown quantities in two separate equations describing the total force. The block in (Figure 1) has a mass m = 10kg and is being pulled by a...
-
Show the following relationships, use indicial notation: (A-a) (B-b) =a (A-B)-b a--b=b. A-a
-
In the middle of next year, ABC Ltd will need to borrow $20m from the market. They are going to issue bills, but are worried that the rates may increase by next year. Explain to ABC Ltd how you can...
-
A college lab does testing for outsiders for a fee of $50 per hour. You have two technicians devoted to this purpose. You know from experience that they have enough work to keep them busy most of the...
-
On April 1, Catherine's employer provided her with a $100,000 loan at an annual interest rate of 5%. Assume that the prescribed interest rate was 3% for the year. If Catherine paid interest on the...
-
3. Explain the following. a) Relationship between risk and rate of returns. b) How do you estimate a firm's cost of new debt financing? c) Does dividend policy affect stock price? Explain
-
Explore the intersection of typography and cognitive psychology, interrogating the perceptual and cognitive mechanisms that underpin the legibility, readability, and typographic processing of textual...
-
The sales forecast for January to May 20x4 and actual sales for November and December 20x3 for Purple Company are as follows: Month Sales Actual (20x3) November ...
-
Vectors are drawn from the center of a regular n-sided polygon in the plane to the vertices of the polygon. Show that the sum of the vectors is zero.
-
Draw two constitutional isomers of cis-1, 2-dibromo-cyclopentane.
-
How would you prepare the following compounds from 2-phenylethanol? More than one step may be required. (a) Styrene (PhCH = CH2 (b) Phenyl acetaldehyde (PhCH2CHO) (c) Phenyl acetic acid (PhCH2CO2H)...
-
The primary fate of acetyl CoA under normal metabolic conditions is degradation in the citric acid cycle to yield CO2. When the body is stressed by prolonged starvation, however, acetyl CoA is...
-
Which of the following provides evidence for evolution? (a) changes in the coloration of peppered moth populations over time (b) the presence of vestigial eyes in cave salamanders (c) the fact that...
-
Which of these adaptations is the result of sexual selection? (a) the spines on cactus plants (b) parental care in male poison dart frogs (c) the songs of male birds (d) dark wings in peppered moths
-
If you put a heavy load in a wheelbarrow, the force you apply to the handles is much less than the weight of the load and the wheelbarrow. To lift with the least force, you put the load toward the...

Study smarter with the SolutionInn App



