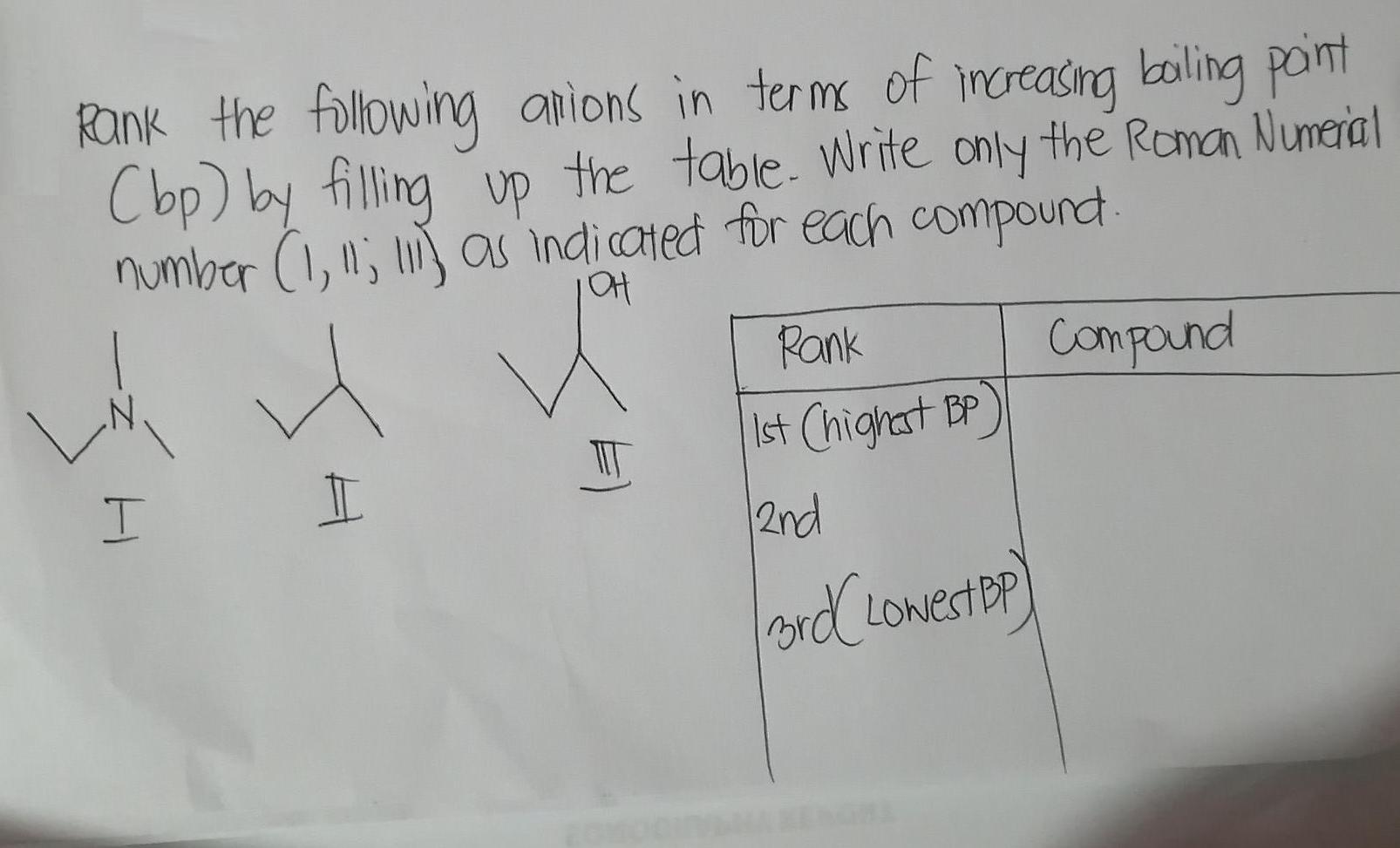
Rank the following anions in terms of imcreasing boling pont Cbp) by filling up the table....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Rank the following anions in terms of imcreasing boling pont Cbp) by filling up the table. Write only the Roman Numeral number (1,li; l} as indicated for each compoundt Pank Compound Ist Chighat BP 2nd Rank the following anions in terms of imcreasing boling pont Cbp) by filling up the table. Write only the Roman Numeral number (1,li; l} as indicated for each compoundt Pank Compound Ist Chighat BP 2nd
Expert Answer:
Answer rating: 100% (QA)
Boiling point of 2butanol is 99 0 Boiling point of NNdimethylethylamine is 365 0C Boiling point of 2... View the full answer

Posted Date:
Students also viewed these chemistry questions
-
Rank the following anions in terms of increasing basicity:
-
Rank the following anions in order of decreasing basicity: CH O CH,o CH
-
Write the numeral as numerals in the indicated systems of numeration. In HinduArabic, Egyptian, Roman, and Greek
-
Expected manufacturing costs for Imperial Data Devices are as follows: Variable Costs Fixed Costs per Month Direct material $8.00/unit Supervisory salaries $17,000 Direct labor 3.50/unit Factory...
-
Multiple Choice Questions 1. The receiving department employs one worker, who spends 25 percent of his time on the receiving activity and 75 percent of his time on inspecting products. His salary is...
-
Presented below are the financial statements of Helwany Company. Additional data:1. Depreciation expense was $17,500.2. Dividends declared and paid were $20,000.3. During the year equipment was sold...
-
Draw a graph similar to the one shown in exhibit 13.4 and explain its implications. Exhibit 13.4 EXHIBIT 13.4 Bayside Memorial Hospital: Corporate and Project Costs of Capital Project Cost of Capital...
-
On April 1, 2012, Dougherty Inc. entered into a cost plus- fixed-fee contract to construct an electric generator for Altom Corporation. At the contract date, Dougherty estimated that it would take 2...
-
19. Consider the given arrangement. The two slits S and S are illuminated by monochromatic light of wavelength A. Slits S3 and S4 are at separation d minimum intensity on the screen will be 2.D =...
-
Write a query to display the patron ID, book number, and days kept for each checkout. Days Kept is the difference from the date on which the book is returned to the date it was checked out. Sort the...
-
Write a paper about knowledge management strategy formulation this project provides an opportunity for you to identify opportunities for knowledge management at a real-world business and to suggest...
-
Mergers have been an important part of the airline industry. Have they been successful? Why have so many been unsuccessful?
-
Why might an airline choose direct flights rather than hub and spoke?
-
With changes in fuel prices, what kind of effects can be witnessed in the market for air travel?
-
Why might an airline that has international routes pay the workers in one country more than the workers in another country for essentially the same job?
-
Do you think that the problem of scarcity will ever be solved? Why or why not?
-
Currently the rate of return of the risk-free government bond is 3% while the expected rate of return on the market is 11%. Using CAPM, calculate the rate of return required for A plc who has a beta...
-
6 (a) Briefly develop a mathematical model of the behaviour of a copper-twisted pair cable (b) Derive the magnetic energy from: w given that: K + w, where the - - k symbols have their usual meaning...
-
You shoot a beam of electrons through a double slit to make an interference pattern. After noting the properties of the pattern, you then double the speed of the electrons. What effect would this...
-
Photon P in Figure Q28.35 moves an electron from energy level \(n=1\) to energy level \(n=3\). The electron jumps down to \(n=2\), emitting photon \(\mathrm{Q}\), and then jumps down to \(n=1\),...
-
Light consisting of \(2.7 \mathrm{eV}\) photons is incident on a piece of potassium, which has a work function of \(2.3 \mathrm{eV}\). What is the maximum kinetic energy of the ejected electrons? A....

Study smarter with the SolutionInn App

