Rank the following anions in terms of increasing basicity:
Question:
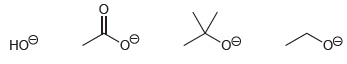
Transcribed Image Text:
ное
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Increa...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Rank the following anions in order of decreasing basicity: CH O CH,o CH
-
Draw all constitutional isomers with molecular formula C 2 H 6 S, and rank them in terms of increasing acidity.
-
For each of the following compounds, rank the highlighted bonds in terms of increasing wave number: a. b. TH. 0= H.
-
Mr. Paulo Rodriguez, a fund manager of The Blackstone Group Inc. holds a portfolio with a current value of RM7.83 million. The cash index currently stands at 1,075 points. He fears that the market...
-
Show that there are inputs that force every percolateDown in heapsort to go all the way to a leaf.
-
The chapter discusses the quest by many consumers to quantify their personal actions exercise, bodily functions, finances, and perhaps even the number of dates they went on this month. As we continue...
-
This exercise considers nursing home data provided by the Wisconsin Department of Health and Family Services (DHFS). Part 1: Use cost-report year 2000 data, and do the following analysis. a....
-
Bayside Coatings Company purchased waterproofing equipment on January 2, 20Y4, for $190,000. The equipment was expected to have a useful life of four years and a residual value of $9,000. Instruction...
-
We are in an incredibly stressful time in our lives. Watch the Ted Talk, "How Burnout Makes Us Less Creative". Share your thoughts on the video. Then discuss what are some actions that you can take...
-
Make-or-buy Beaus Bistro has a reputation for providing good value for its menu prices. The desserts, developed by the pastry chef, are one of the distinctive features of the menu. The pastry chef...
-
Which of the following is not one of the four major economic flows linking the U.S. economy with that of other nations? a. Trade flows. b. Resource flows. c. Financial flows. d. Foreign aid flows.
-
Compare a hypothetical DVC with a hypothetical IAC. In the DVC, average per capita income is $500 per year. In the IAC, average per capita income is $40,000 per year. If both countries have a savings...
-
In Problem graph each of the functions using the graphs of functions f and g below. y = f(x + 2) f(x) g(x) -5 -5 5 -5 -5
-
Have you ever been misinterpreted in an email or instant message because the receiver could not see your facial expressions or hear your tone of voice? Describe what happened and how you handled the...
-
How to improve vaccination rates has been a publicized topic over the last few years. There is controversy on the safety and unknown adverse negative externalities. A lack of trust in our government,...
-
What are the potential downsides of authoritarian, democratic, and laissez-faire styles of leadership? When is laissez-faire leadership appropriate? Why? What style of leadership (authoritarian,...
-
What is the maximum capacity utilization rating a company should strive for with no side effects? Explain.
-
If you were the leader of your organization, how would you change the way your organization communicates in order to assure that all employees contribute to the organization's mission, vision, and...
-
Determine whether the graph shown is connected or disconnected. B G
-
Distinguish between the work performed by public accountants and the work performed by accountants in commerce and industry and in not-for-profit organisations.
-
Tell the number of hydrogens bonded to each carbon atom in the following substances and give the molecular formula ofeach: OH H (a) (b) CO2CH3 Ephedrine Cocaine
-
Identify the most electronegative element in each of the following molecules: (a) CH2FC1 (b) FCH2CH2CH2Br (c) HOCH2CH2NH2 (d) CH3OCH2Li
-
Use the electro negatively table (Figure) to predict which bond in each of the following sets is more polar, and indicate the direction of bond polarity for each compound. (a) H3C ? C1 OR C1 ? C1 (b)...
-
Using Fourier Transforms, solve the following integral Equation for ((t) f (t) - ( ( ( t -u) ue - 2 u H (u) du = foo_flt -4t e H(t)
-
Q1: what is the net operating income under the variable and absorption cost system. A: ACER company Product sales: 1,000 units at $10 each Variable manufacturing costs: $5.50 per unit Fixed...
-
Solve for 2 and graph the solution on the number line below. 245x+9 or 5x +9 69

Study smarter with the SolutionInn App


